A dihydrogen molecule (H2) forms from two hydrogen atoms. When the atomic orbitals of the two atoms combine, the electrons occupy the molecular orbital of lowest energy, the σ1s bonding orbital. A dihydrogen molecule, H2, readily forms because the energy of a H2 molecule is lower than that of two H atoms. The σ1s orbital that contains both electrons is lower in energy than either of the two 1s atomic orbitals.
A molecular orbital can hold two electrons, so both electrons in the H2 molecule are in the σ1s bonding orbital; the electron configuration is $(σ_{1s})^2$. We represent this configuration by a molecular orbital energy diagram (below) in which a single upward arrow indicates one electron in an orbital, and two (upward and downward) arrows indicate two electrons of opposite spin.
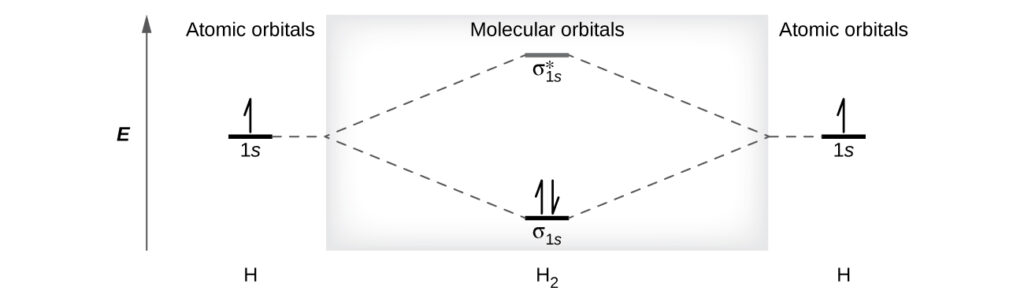
A dihydrogen molecule contains two bonding electrons and no antibonding electrons so we have
$$\text{bond order in }H_2=\frac{(2−0)}{2}=1$$
Because the bond order for the H–H bond is equal to 1, the bond is a single bond.
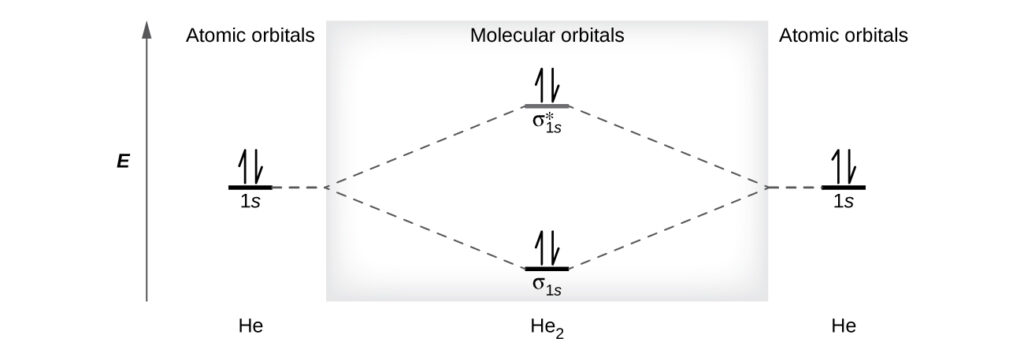
A helium atom has two electrons, both of which are in its 1s orbital. Two helium atoms do not combine to form a dihelium molecule, He2, with four electrons, because the stabilizing effect of the two electrons in the lower-energy bonding orbital would be offset by the destabilizing effect of the two electrons in the higher-energy antibonding molecular orbital. We would write the hypothetical electron configuration of He2 as $(σ_{1s})^2(σ_{1s}^*)^2$ . The net energy change would be zero, so there is no driving force for helium atoms to form the diatomic molecule. In fact, helium exists as discrete atoms rather than as diatomic molecules. The bond order in a hypothetical dihelium molecule would be zero.
$$\text{bond order in }He_2=\frac{(2−2)}{2}=0$$
A bond order of zero indicates that no bond is formed between two atoms.