Calculating values for equilibrium constants and/or equilibrium concentrations is of practical benefit to many applications. A mathematical strategy that uses initial concentrations, changes in concentrations, and equilibrium concentrations (and goes by the acronym ICE) is useful for several types of equilibrium calculations.
Practice Problems: Equilibrium Calculations
Calculating K values from equilibrium states
An equilibrium mixture of NH3(g), H2(g), and N2(g) at 500 °C was found to contain 1.35 M H2, 1.15 M N2, and $4.12×10^{-1}$ M NH3.
What is the value of the equilibrium constant at 500 °C for the formation of NH3 according to the following equation?
$$N_2(g)+3\, H_2(g) \rightleftharpoons 2\, NH_3(g)$$
Solution
$$K_C = \frac{[NH_3 ]^2}{[N_2][H_2]^3}\\ K_C = \frac{[4.12×10^{-1}\, \text{M}]^2}{[1.15\, \text{M}][1.35\, \text{M}]^3}\\ \mathbf{K_C = 6.00×10^{-2}}$$
Hydrogen is prepared commercially by the reaction of methane and water vapor at elevated temperatures.
$$CH_4(g)+H_2O(g) \rightleftharpoons 3\, H_2(g)+CO(g)$$
What is the equilibrium constant for the reaction if a mixture at equilibrium contains gases with the following concentrations: CH4, 0.126 M; H2O, 0.242 M; CO, 0.126 M; H2 1.15 M, at a temperature of 760 °C?
Solution
$$K_C = \frac{[H_2 ]^3[CO]}{[CH_4][H_2 O]}\\ K_C = \frac{[1.15\, \text{M}]^3 [0.126\, \text{M}]}{[0.126\, \text{M}][0.242, \text{M}]}\\ \mathbf{K_C = 6.28}$$
A 0.72-mol sample of PCl5 is put into a 1.00-L vessel and heated. At equilibrium, the vessel contains 0.40 mol of PCl3(g) and 0.40 mol of Cl2(g). Calculate the value of the equilibrium constant for the decomposition of PCl5 to PCl3 and Cl2 at this temperature.
Solution
$$PCl_5 \rightleftharpoons PCl_3 + Cl_2 \\$$
| $PCl_5$ | $\rightleftharpoons $ | $PCl_3$ | + | $Cl_2$ | |
| Initial Concentrations | 0.72 M | 0 | 0 | ||
| Change in Concentrations | -x | +x | +x | ||
| Equilibrium Concentrations | 0.72-x =0.72 – 0.40 M = 0.32 M |
x =0.40 M |
x =0.40 M |
At 1 bar and 25 °C, NO2 with an initial concentration of 1.00 M is 0.0033% decomposed into NO and O2. Calculate the value of the equilibrium constant for the reaction. $$2\,NO_2(g) \rightleftharpoons 2\,NO(g)+O_2(g)$$
Solution
0.0033% decomposed means that 99.9967% remains as NO2: $[NO_2] = 99.9967\% \times 1.00\, \text{M} = 0.999_{967}\, \text{M}$.
| $2\,NO_2$ | $\rightleftharpoons $ | $2\,NO$ | + | $O_2$ | |
| Initial Concentrations | 1.00 M | 0 | 0 | ||
| Change in Concentrations | -2x | +2x | +x | ||
| Equilibrium Concentrations | 1.00-2x =1.00 – 0.000033 M = $0.999_{967}\, \text{M}$ |
2x =(0.000033) M = $3.3 \times 10^{-5}\, \text{M}$ |
x =$\frac{0.000033}{2} M$ = $1.65\times 10^{-5}\, \text{M}$ |
Calculate the value of the equilibrium constant KP for the reaction $2\,NO(g)+Cl_2(g) \rightleftharpoons 2\,NOCl(g)$ from these equilibrium pressures: NO, 0.050 bar; Cl2, 0.30 bar; NOCl, 1.2 bar.
Solution
$$K_P = \frac{(P_{NOCl})^2}{(P_{NO})^2 (P_{Cl_2})}\\ K_P = \frac{(1.2\; \text{bar})^2}{(0.050\; \text{bar})^2(0.30\; \text{bar})}\\ \mathbf{K_P = 1.9×10^3}$$
When heated, iodine vapor dissociates according to this equation:
$$I_2(g) \rightleftharpoons 2I(g)$$
At 1274 K, a sample exhibits a partial pressure of I2 of 0.1122 bar and a partial pressure due to I atoms of 0.1378 bar. Determine the value of the equilibrium constant, KP, for the decomposition at 1274 K.
Solution
$$K_P = \frac{(P_I)^2}{P_{I_2}}\\ K_P = \frac{0.1378^2}{0.1122} \\ K_P = 0.1692_{40998} \\ \mathbf{K_P = 0.1692}$$
A sample of ammonium chloride was heated in a closed container. $$NH_4Cl(s) \rightleftharpoons NH_3(g)+HCl(g)$$ At equilibrium, the pressure of NH3(g) was found to be 1.75 bar. What is the value of the equilibrium constant KP for the decomposition at this temperature?
Solution
The amount of $NH_3$ produced is equal to the amount of $HCl$ produced in this reaction. Since we started with only annominum chloride in the container, the equilibrium pressures of the products must be equal. $$K_P = (P_{NH_3})(P_{HCl}) \\ K_P = (1.75\, \text{bar})(1.75\, \text{bar}) \\ K_P = 3.06_{25} \\ \mathbf{K_P = 3.06}$$
At a temperature of 60 °C, the vapor pressure of water is 0.196 bar. What is the value of the equilibrium constant KP for the vaporization equilibrium at 60 °C?
$$H_2O(l) \rightleftharpoons H_2O(g)$$
Solution
$$K_P = (P_{H_2O}) \\ K_P = (0.196\, \text{bar}) \\ \mathbf{K_P = 0.196}$$
In the ICE table in the solution for the example here, the initial and final values for water are not recorded, but a change is listed. Explain what this notation means: why concnetrations are not recorded, and what is the implication of including the “change” row.
Solution
The water in this reaction is a pure liquid – it does not have a defined concentration (or: its activity is a constant at 1). Therefore having an initial or final value for “concentration” would be meaningless for this species. However, the amount of water can change during the reaction (the number of moles present) can change during the reaction. Including the stoichiometry in the Change row can help track whether the amount of water should increase or decrease – and if we knew the intial amount (in mol or g), we could calculate the final amount (in mol or g) based on the reaction stoichiometry.
Calculations with missing equilibrium concentrations
Analysis of the gases in a sealed reaction vessel containing NH3, N2, and H2 at equilibrium at 400 °C established the concentration of N2 to be 1.2 M and the concentration of H2 to be 0.24 M.
$$N_2(g)+3H_2(g) \rightleftharpoons 2NH_3(g)\qquad K_c=0.50\;\text{at 400°C}$$Calculate the equilibrium molar concentration of NH3.
Solution
$$K_C = \frac{[NH_3]^2}{[N_2][H_2]^3} \\ 0.50 = \frac{[NH_3]^2}{[1.2\, \text{M}][0.24\, \text{M}]^3} \\ [NH_3] = 0.091_{0736} \, \text{M}\\ \mathbf{[NH_3] = 0.091 \, \text{M}}$$
Calculate the number of moles of HI that are at equilibrium with 1.25 mol of H2 and 1.25 mol of I2 in a 5.00−L flask at 448 °C.
$$H_2+I_2 \rightleftharpoons 2HI\qquad K_c=50.2\;\text{at 448°C}$$Solution
$$ K_C = \frac{[HI]^2}{[H_2][I_2]}\\ 50.2 = \frac{[HI]^2}{[\frac{1.25\, \text{mol}}{5.00\, \text{L}}][\frac{1.25\, \text{mol}}{5.00\, \text{L}}]} \\ [HI] = 1.77_{1298}\, \text{M}\\ \text{mol HI} = 1.77_{1298}\, \text{M} \times 5.00\, \text{L} \\ \text{mol HI} = 8.85_{64947}\, \text{mol} \\ \mathbf{\text{mol HI} = 8.86\, \text{mol}} $$
What is the pressure of BrCl in a mixture of Cl2, Br2, and BrCl at equilibrium if the pressure of Cl2 in the mixture is 0.115 bar and the pressure of Br2 in the mixture is 0.450 bar?
$$Cl_2(g)+Br_2(g) \rightleftharpoons 2BrCl(g)\qquad K_P=4.7×10^{-2}$$Solution
$$K_P = \frac{(P_{BrCl})^2}{(P_{Cl_2})(P_{Br_2})}\\ 4.7×10^{-2} = \frac{(P_{BrCl})^2}{(0.115\, \text{bar})(0.450\, \text{bar})} \\ P_{BrCl} = 0.049_{317846}\, \text{bar} \\ \mathbf{P_{BrCl} = 0.049\, \text{bar}}$$
What is the pressure of CO2 in a mixture that contains 0.50 bar H2, 2.0 bar of H2O, and 1.0 bar of CO at equilibrium at 990 °C?
$$H_2(g)+CO_2(g) \rightleftharpoons H_2O(g)+CO(g)\qquad K_P=1.6\;\text{at 990°C}$$Solution
$$K_P = \frac{(P_{H_2 O})(P_{CO})}{(P_{H_2})(P_{CO_2})} \\ 1.6 = \frac{(2.0\, \text{bar})(1.0\, \text{bar})}{(0.50\, \text{bar})(P_{CO_2})} \\ \mathbf{P_{CO_2} = 2.5\, \text {bar}}\\$$
Cobalt metal can be prepared by reducing cobalt(II) oxide with carbon monoxide.
$$CoO(s)+CO(g) \rightleftharpoons Co (s) +CO_2(g)\qquad K_c=4.90×10^2\;\text{at 550°C}$$If a reaction vessel was initially prepared containing only CoO and CO, what concentration of CO remains in an equilibrium mixture with [CO2] = 0.100 M?
Solution
$$K_C = \frac{[CO_2]}{[CO]}\\ 4.90\times 10^2 = \frac{[0.100\, \text{M}]}{[CO]} \\ [CO] = 2.04_{0816327}\, \times 10^{-4}\, \text{M}\\ \mathbf{[CO] = 2.04\, \times 10^{-4}\, \text{M}}\\$$
Carbon reacts with water vapor at elevated temperatures. $$C(s)+H_2O(g) \rightleftharpoons CO(g)+H_2(g)\qquad K_c=0.2\;\text{at 1000°C}$$ Assuming a reaction mixture initially contains only reactants, what is the concentration of CO in an equilibrium mixture with [H2O] = 0.500 M at 1000 °C?
Solution
Since the only source of CO and H2 is this reaction and both are produced in equal amounts, [CO] = [H2]. $$K_C = \frac{[CO][H_2]}{[H_2 O]}\\ 0.2 = \frac{[CO]^2}{[0.500\, \text{M}]}\\ [CO] = 0.3_{16227766}\, \text{M}\\ \mathbf{[CO] = 0.3\, \text{M}}$$
Sodium sulfate decahydrate, Na2SO4·10H2O, dehydrates according to the reaction $$Na_2SO_4\cdot 10\, H_2O(s) \rightleftharpoons Na_2SO_4(s)+10\, H_2O(g)\qquad K_P=4.08×10^{-25}\;\text{at 25°C}$$ What is the pressure of water vapor at equilibrium with a mixture of Na2SO4·10H2O and NaSO4?
Solution
$$K_P = (P_{H_2 O})^{10}\\ 4.08\times 10^{-25} = (P_{H_2 O})^{10}\\ P_{H_2O} = 3.63_{970} \times 10^{-3}\, \text{bar}\\ \mathbf{P_{H_2O} = 3.64 \times 10^{-3}\, \text{bar}}$$ **Hint: to do a 10th root on your calculator, you can either use the $\sqrt[x]\quad $ key (i.e. enter $\require{enclose} \enclose{box}{10}\; \enclose{box}{\sqrt[x]\quad}\; \enclose{box}{5}$ to take the 10th root of 5) OR raise your value to a fractional power ( $ 5^{\frac{1}{10}} = 5^{0.1}$ is equivalent to $\sqrt[10]{5} $).
Calcium chloride hexahydrate, CaCl2·6H2O, dehydrates according to the equation $$CaCl_2\cdot 6\, H_2O(s) \rightleftharpoons CaCl_2(s)+6\, H_2O(g)\qquad K_P=5.09×10^{-44}\;\text{at 25°C}$$ What is the pressure of water vapor at equilibrium with a mixture of CaCl2·6H2O and CaCl2 at 25 °C?
Solution
$$K_P = (P_{H_2O})^6\\ 5.09×10^{-44} = (P_{H_2O})^6\\ P_{H_2O} = 6.08_{7696}\times 10^{-8}\, \text{bar}\\ \mathbf{P_{H_2O} = 6.09\times 10^{-8}\, \text{bar}} $$ (see note in previous question on calculating higher roots on your calculator)
A student solved the following problem (incorrectly) and found [N2O4] = 0.16 M at equilibrium. How could this student recognize that the answer was wrong without reworking the problem? The problem was:
What is the equilibrium concentration of N2O4 in a mixture formed from a sample of NO2 with a concentration of 0.10 M? $$2NO_2(g) \rightleftharpoons N_2O_4(g)\qquad K_c=160$$
Solution
The student could check their value by solving for K using their calculated value: $$K_C = \frac{[N_2 O_4]}{[NO_2]^2}\\ K_C = \frac{[0.16\, \text{M}]}{[0.10\, \text{M}]^2}\\ K_C = 16 \neq 160$$ Since the K determined with the calculated concentrations does not equal the given K, an error in calculation must have been made when finding [N2O4].
Calculations with ICE tables
Calculate the equilibrium concentration of both species in 1.00 L of a solution prepared from 0.129 mol of N2O4 with chloroform as the solvent. $$N_2O_4\,(g) \rightleftharpoons 2\,NO_2\,(g)\qquad K_c=1.07×10^{-5}\;\text{in chloroform}$$
Solution
The reaction as intially prepared has 0 NO2, so it can only proceed in the forward direction (right / towards the products):
| $N_2O_4\,(g)$ | $\rightleftharpoons $ | $2\,NO_2\,(g) $ | |
| Initial Concentrations: | $\frac{0.129\, \text{mol}}{1.00\, \text{L}}$ | $0$ | |
| Change in Concentration: | -x | +2x | |
| Equilibrium Concentrations: | $0.129-x\,\text{M} \\ \approx 0.129\, \text{M}$ | $2x\,\text{M} \\ \; \;$ |
Calculate the equilibrium concentration of all species in an equilibrium mixture that results from the decomposition of COCl2 with an initial concentration of 0.3166 M. $$COCl_2\,(g) \rightleftharpoons CO\, (g)+Cl_2\,(g)\qquad K_c=2.2×10^{-10}$$
Solution
The initial mixture is prepared with only COCl2; the reaction can only proceed forward (right / towards products):
| $COCl_2\,(g)$ | $\rightleftharpoons $ | $CO\, (g) $ | + | $Cl_2\,(g)$ | |
| Initial Concentrations: | 0.3166 M | 0 | 0 | ||
| Change in Concentration: | -x | +x | +x | ||
| Equilibrium Concentrations: | 0.3166 – x $\approx$ 0.3166 M |
x | x |
Calculate the equilibrium pressures of all species in an equilibrium mixture that results from the decomposition of H2S with an initial pressure of 0.824 bar. $$2H_2S(g) \rightleftharpoons 2H_2(g)+S_2(g)\qquad K_P=2.2×10^{-6}$$
Solution
The initial mixture is prepared with only H2S; the reaction can only proceed forward (right / towards products):
| $2\, H_2 S\,(g)$ | $\rightleftharpoons $ | $2\, H_2\, (g) $ | + | $S_2\,(g)$ | |
| Initial Pressures: | 0.824 bar | 0 | 0 | ||
| Change in Pressure: | -2x | +2x | +x | ||
| Equilibrium Pressures: | 0.824 – 2x $\approx$ 0.924 bar |
2x | x |
What are all concentrations after a mixture that contains [H2O] = 1.00 M and [Cl2O] = 1.00 M comes to equilibrium at 25 °C? $$H_2O(g)+Cl_2O(g) \rightleftharpoons 2HOCl(g)\qquad K_c=0.0900$$
Solution
The initial mixture is prepared with only reactants; the reaction can only proceed forward (right / towards products):
| $H_2 O\,(g)$ | + | $Cl_2 O\,(g)$ | $\rightleftharpoons $ | $2\, HOCl\, (g) $ | |
| Initial Concentrations: | 1.00 M | 1.00 M | 0 | ||
| Change in Concentration: | -x | -x | +2x | ||
| Equilibrium Concentration: | 1.00 – x | 1.00 – x | 2x |
What are the concentrations of PCl5, PCl3, and Cl2 in an equilibrium mixture produced by the decomposition of a sample of pure PCl5 with [PCl5] = 2.00 M? $$PCl_5(g) \rightleftharpoons PCl_3(g)+Cl_2(g)\qquad K_c=0.0211$$
Solution
The initial mixture is prepared with only PCl5; the reaction can only proceed forward (right / towards products):
| $PCl_5\, (g)$ | $\rightleftharpoons $ | $PCl_3\, (g) $ | + | $Cl_2\,(g)$ | |
| Initial Concentrations: | 2.00 M | 0 | 0 | ||
| Change in Concentration: | -x | +x | +x | ||
| Equilibrium Concentration: | 2.00 – x | x | x |
Calculate the number of grams of HI that are at equilibrium in a container initially prepared with 1.25 mol of H2 and 63.5 g of iodine in a 2.00 L container at 448 °C. $$H_2\, (g) +I_2\, (g) \rightleftharpoons 2HI\, (g)\qquad K_c=50.2\;\text{at 448°C}$$
Solution
The initial mixture is prepared with only reactants; the reaction can only proceed forward (right / towards products).
The initial amounts of the reagents are provided in mol and g rather than concentration; they should be converted to concentration before use in the calculation for K.
| $H_2\, (g)$ | + | $I_2\,(g)$ | $\rightleftharpoons $ | $2\, HI\, (g) $ | |
| Initial Concentrations: | $\frac{1.25\, \text{mol}}{2.00\, \text{L}}\\ = 0.625\, \text{M}$ | $\frac{63.5\, \text {g}\frac{1\, \text{mol}\, I_2}{253.8\, \text{g}}}{2.00\, \text{L}}\\ = 0.125_{0985} \, \text{M}$ | 0 | ||
| Change in Concentration: | -x | -x | +2x | ||
| Equilibrium Concentration: | 0.625 – x | 0.1250985 – x | 2x |
Butane exists as two isomers, n−butane and isobutane.
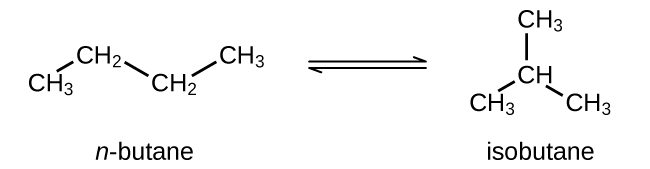
KP for the reaction above is 2.5 at 25 °C
What is the pressure of isobutane in a container of the two isomers at equilibrium with a total pressure of 1.22 bar?
Solution
The total pressure in the system is the sum of the partial pressures of all gases present in the mixture: $$P_{total} = 1.22\, \text{bar} = P_{n-\text{butane}} + P_{\text{isobutane}} $$ And we know the expression for $K_P$ from the reaction given: $$K_P = 2.5 = \frac{P_{\text{isobutane}}}{P_{n-\text{butane}}}$$ We can rearrange the expression for $K_P$ to solve for $P_{\text{isobutane}}$ and substitute this into the sum of pressures above: $$2.5\cdot P_{n- \text{butane}} = P_{\text{isobutane}} \\ 1.22\, \text{bar} = P_{n-\text{butane}} + P_{\text{isobutane}} \\ 1.22\, \text{bar} = P_{n-\text{butane}} + (2.5\cdot P_{n-\text{butane}}) \\ 1.22\, \text{bar} = 3.5\cdot P_{n-\text{butane}} \\ \mathbf{P_{n-\text{butane}}} = 0.34_{85714}\, \text{bar} = \mathbf{0.35\, \text{bar}} \\ 2.5\cdot P_{n-\text{butane}} = P_{\text{isobutane}}\\ 2.5 (0.34_{85714}\, \text{bar}) = P_{\text{isobutane}}\\ \mathbf{P_{\text{isobutane}}} = 0.87_{142857}\, \text{bar} = \mathbf{0.87\, \text{bar}}$$ As a check, we can verify that the total pressure and K values are still valid: $$P_{total} = 0.87\, \text{bar} + 0.35\, \text{bar} = 1.22\, \text{bar}\ ✓ \\ K = \frac{0.87\, \text{bar}}{0.35\, \text{bar}} = 2.5\ ✓ $$
What is the minimum mass of CaCO3 required to establish equilibrium at a certain temperature in a 6.50-L container if the equilibrium constant (Kc) is 0.50 for the decomposition reaction of CaCO3 at that temperature?
$$CaCO_3(s) \rightleftharpoons CaO(s)+CO_2(g)$$Solution
Remember – although a solid does not appear in the expression for $K$, its presence is required in order to reach equilibrium!
| $CaCO_3\, (s)$ | $\rightleftharpoons $ | $CaO\, (s) $ | + | $CO_2(g)$ | |
| Initial Concentrations: | $\require{cancel} \cancel{\;\;\;\;}$ | $\require{cancel} \cancel{\;\;\;\;}$ | 0 | ||
| Change in Concentrations: | -x | +x | +x | ||
| Equilibrium Concentrations: | $\require{cancel} \cancel{\;\;\;\;}$ | $\require{cancel} \cancel{\;\;\;\;}$ | x |
Setting up the ICE table with the “change” in the solids visible highlights that we need at least “x” amount of the CaCO3 to exist in the container or else we will have a negative amount at equilibrium (not possible – the reaction will be unable to reach equilibrium). Solving for “x” and conveting to mass of CaCO3: $$K_C = [CO_2] \\ 0.50 \, \text{M} = x \\ 0.50 \, \text{M} \times 6.50\, \text{L} \times \frac{100.1\, g\, CaCO_3}{\text{mol}} \\ = 32_{5.325} \, \text{g}\, CaCO_3 \\ \mathbf =3.3\times 10^2\, \text{g}\, CaCO_3$$
The equilibrium constant (Kc) for this reaction is 1.60 at 990 °C:
$$H_2(g)+CO_2(g) \rightleftharpoons H_2O(g)+CO(g)$$Calculate the number of moles of each component in the final equilibrium mixture obtained from adding 1.00 mol of H2, 2.00 mol of CO2, 0.750 mol of H2O, and 1.00 mol of CO to a 5.00-L container at 990 °C.
Solution
This reaction starts with some of each reagent present: in order to determine the direction in which the reaction will proceed, we should compare the reaction quotient Q to the equilibrium constant: $$Q_C = \frac{[H_2 O][CO]}{[H_2][CO_2]}\\ Q_C = \frac{\left[ \frac{0.750\, \text{mol}}{5.00\, \text{L}} \right] \left[ \frac{1.00\, \text{mol}}{5.00\, \text{L}} \right]}{\left[ \frac{1.00\, \text{mol}}{5.00\, \text{L}} \right] \left[ \frac{2.00\, \text{mol}}{5.00\, \text{L}} \right]} \\ Q_C = 0.375 \lt K_C (1.60) $$ The reaction will proceed forward (towards the products).
| $H_2\, (g)$ | + | $CO_2(g)$ | $\rightleftharpoons $ | $H_2 O (g) $ | + | $CO(g)$ | |
| Initial Concentrations: | $\frac{1.00\, \text{mol}}{5.00\, \text{L}} \\ = 0.200\, \text {M}$ | $\frac{2.00\, \text{mol}}{5.00\, \text{L}} \\ = 0.400\, \text {M}$ | $\frac{0.750\, \text{mol}}{5.00\, \text{L}} \\ = 0.150\, \text {M}$ | $\frac{1.00\, \text{mol}}{5.00\, \text{L}} \\ = 0.200\, \text {M}$ | |||
| Change in Concentrations: | -x | -x | +x | +x | |||
| Equilibrium Concentrations: | 0.200 – x | 0.400 – x | 0.150 + x | 0.200 + x |
Since no initial concentration is large compared to the others, it is not reasonable to use the “small x” approximation here (if you ignored one $x$, all would be ignored, creating an unsolvable situation). Solve for $x$ algebraically using the quadratic formula: $$K_C = \frac{[H_2 O][CO]}{[H_2][CO_2]}\\ 1.60 = \frac{[0.150 + x][0.200 + x]}{[0.200 – x][0.400 – x]} \\ 0 = 0.600x^2 – 1.31x + 0.098\\ \quad \text{Solve with the quadratic formula and a = +0.600, b= – 1.31, c = + 0.098 :} \\ x = \frac{-b\pm \sqrt{b^2- 4ac}}{2a} \\ x = \frac{-(- 1.31) \pm \sqrt{(- 1.31)^2 – 4(0.600)(0.098)}}{2(0.600)} \\ x = 0.07756471, 2.1057686 \\ \quad \text{Only the first root makes chemical sense; the other will create (impossible) negative concentrations.}\\ x = 0.0775_{6471} \\ [H_2] = 0.200 – x = 0.200 – 0.0775_{6471} = 0.122_{435} \, \text{M} \times 5.00\, \text{L} = 0.612_{176}\, \text{mol} = \mathbf{0.612\, \text{mol}\, H_2}\\ [CO_2] = 0.400 – x = 0.400 – 0.0775_{6471} = 0.322_{435} \, \text{M} \times 5.00\, \text{L} = 1.61_{2176}\, \text{mol} = \mathbf{1.61\, \text{mol}\, CO_2}\\ [H_2 O] = 0.150 + x = 0.150 + 0.0775_{6471} = 0.227{5647} \, \text{M} \times 5.00\, \text{L} = 1.13_{782}\, \text{mol} = \mathbf{1.14\, \text{mol}\, H_2 O}\\ [H_2] = 0.200 + x = 0.200 + 0.0775_{6471} = 0.277_{5647} \, \text{M} \times 5.00\, \text{L} = 1.38_{782}\, \text{mol} = \mathbf{1.38\, \text{mol}\, H_2}$$
In a 3.0-L vessel, the following equilibrium partial pressures are measured: N2, 0.190 bar; H2, 0.317 bar; NH3, 1.00 bar.
$$N_2(g)+3\, H_2(g) \rightleftharpoons 2\, NH_3(g)$$(a) How will the partial pressures of H2, N2, and NH3 change if H2 is removed from the system? Will they increase, decrease, or remain the same?
Solution
Since H2 is a reactant in this system, removing it will cause the equilibrium to shift left (towards the reactants). The partial pressure of ammonia will decrease, while the partial pressure of N2 will increase. The partial pressure of H2 will also increase, but its new equilibrium pressure will be lower than the initial 0.317 bar since some was removed.
(b) Hydrogen is removed from the vessel until the partial pressure of nitrogen, at equilibrium, is 0.250 bar. Calculate the partial pressures of the other substances under the new conditions.
Solution
The partial pressure of hydrogen is being manipulated by the experimenter, so its value will not follow stoichiometry. All the other products and reactants will maintain their stoichiometric ratios however. Since the partial pressure of N2 increased from 0.190 bar to 0.250 bar, the change in pressure during the manipulation is $(0.250 – 0.190) = -0.060$ bar. This value is analagous to the “+x” that would appear in the Change row in an ICE table for the reaction proceeding “left”.
If nitrogen pressure increased by “+x”, the ammonia pressure should decrease by double that value (“-2x”): $\mathbf{P_{NH_3}} = 1.00 \, \text{bar} – 2(0.060) = \mathbf{0.88}$ bar.
Since we don’t know how much hydrogen gas was removed, we cannot calculate its new equilibrium pressure stoichiometrically, but we can find its equilibrium pressure from the K value and two known pressures. We are not told the K value directly, but we can find it from the first set of equilibrium pressures:
$$K_P = \frac{(P_{NH_3})^2}{(P_{N_2})(P_{H_2})^3} \\
K_P = \frac{(1.00)^2}{(0.19)(0.317)^3} \\
K_P = 165_{.222}$$
Now we can use this K value to solve the new equilibrium:
$$K_P = \frac{(P_{NH_3})^2}{(P_{N_2})(P_{H_2})^3} \\
165_{.222} = \frac{(0.88)^2}{(0.250)(P_{H_2})^3} \\
P_{H_2} = 0.26_{5655} \, \text{bar} \\
\mathbf{P_{H_2} = 0.27\, \text{bar}} $$
The equilibrium constant (Kc) for this reaction is 5.0 at a given temperature.
$$CO(g)+H_2O(g) \rightleftharpoons CO_2(g)+H_2(g)$$(a) On analysis, an equilibrium mixture of the substances present at the given temperature was found to contain 0.20 mol of CO, 0.30 mol of water vapor, and 0.90 mol of H2 in 1.00 liters total volume. How many moles of CO2 were there in the equilibrium mixture?
Solution
$$K_C = \frac{[CO_2][H_2]}{[CO][H_2 O]} \\ 5.0 = \frac{[CO_2][0.90\, \text{M}]}{[0.20\, \text{M}][0.30\, \text{M}]} \\ [CO_2] = 0.33_{33333}\, \text{M} \times 1.00\, \text{L} = \mathbf{0.33\, \text{mol}\, CO_2}$$
(b) Maintaining the same temperature, additional H2 was added to the system, and some water vapor was removed by drying. A new equilibrium mixture was thereby established containing 0.40 mol of CO, 0.30 mol of water vapor, and 1.2 mol of H2 in a liter. How many moles of CO2 were in the new equilibrium mixture?
Solution
$$K_C = \frac{[CO_2][H_2]}{[CO][H_2 O]} \\ 5.0 = \frac{[CO_2][1.2\, \text{M}]}{[0.40\, \text{M}][0.30\, \text{M}]} \\ [CO_2] = 0.50\, \text{M} \times 1.00\, \text{L} = \mathbf{0.50\, \text{mol}\, CO_2}$$
(c) Compare this with the quantity in part (a), and discuss whether the second value is reasonable. Explain how it is possible for the water vapor concentration to be the same in the two equilibrium solutions even though some vapor was removed before the second equilibrium was established.
Solution
Since some water vapour was removed, the final water vapour concentration is unlikely to be higher than the starting value once it reaches the new equilibrium – the reaction does not ‘recover’ more than it lost. If removing water vapour was the only change made, the new equilibrium concentration would certainly be lower than the starting value. Since H2 was also added to the system, the reaction is pushed even more to the left while re-establishing equilibrium, further increasing the final water vapour concentration, and allowing it to reach up to the initial value. (Potentially, you could increase the water vapour concentration higher than the starting value by adding even more hydrogen or carbon dioxide gas).
Antimony pentachloride decomposes according to this equation:
$$SbCl_5(g) \rightleftharpoons SbCl_3(g)+Cl_2(g)$$An equilibrium mixture in a 5.00-L flask at 448 °C contains 3.85 g of SbCl5, 9.14 g of SbCl3, and 2.84 g of Cl2. How many grams of each will be found if the mixture is transferred into a 3.00-L flask at the same temperature?
Solution
Initial concentrations: $$[SbCl_5] = \frac{3.85\, \text{g}\, SbCl_5}{5.00\, \text{L}} \frac {1\, \text {mol}}{299.0_5 \, \text{g}\, SbCl_5} = 2.57_{482}\times 10^{-3}\, \text{M}\, SbCl_5\\ [SbCl_3] = \frac{9.14\, \text{g}\, SbCl_5}{5.00\, \text{L}} \frac {1\, \text {mol}}{228.1_5 \, \text{g}\, SbCl_3} = 8.01_{227}\times 10^{-3}\, \text{M}\, SbCl_3\\ [Cl_2] = \frac{2.84\, \text{g}\, SbCl_5}{5.00\, \text{L}} \frac {1\, \text {mol}}{70.90 \, \text{g}\, Cl_2} = 8.01_{128}\times 10^{-3}\, \text{M}\, Cl_2$$ Find $K_C$: $$K_C = \frac{[SbCl_3][Cl_2]}{[SbCl_5]}\\ K_C = \frac{[8.01_{227}\times 10^{-3}\, \text{M}][8.01_{128}\times 10^{-3}\, \text{M}]}{[2.57_{482}\times 10^{-3}\, \text{M}]}\\ K_C = 0.0249_{293}$$ Concentrations on transfer to new container (before any reaction has occurred): $$[SbCl_5] = \frac{3.85\, \text{g}\, SbCl_5}{3.00\, \text{L}} \frac {1\, \text {mol}}{299.0_5 \, \text{g}\, SbCl_5} = 4.29_{136}\times 10^{-3}\, \text{M}\, SbCl_5\\ [SbCl_3] = \frac{9.14\, \text{g}\, SbCl_5}{3.00\, \text{L}} \frac {1\, \text {mol}}{228.1_5 \, \text{g}\, SbCl_3} = 1.33_{537}\times 10^{-2}\, \text{M}\, SbCl_3\\ [Cl_2] = \frac{2.84\, \text{g}\, SbCl_5}{3.00\, \text{L}} \frac {1\, \text {mol}}{70.90 \, \text{g}\, Cl_2} = 1.33_{521}\times 10^{-2}\, \text{M}\, Cl_2$$ Reaction quotient Q: $$Q_C = \frac{[SbCl_3][Cl_2]}{[SbCl_5]}\\ Q_C = \frac{[1.33_{537}\times 10^{-2}\, \text{M}][1.33_{521}\times 10^{-2}\, \text{M}]}{[4.29_{136}\times 10^{-3}\, \text{M}]}\\ Q_C = 0.0415_{485}$$ Q > K: the reaction will proceed in reverse (left, towards the reactant) in order to reach equilibrium.
| $SbCl_5\, (g)$ | $\rightleftharpoons $ | $SbCl_3\, (g) $ | + | $Cl_2\, (g)$ | |
| Initial Concentrations: | $4.29_{136}\times 10^{-3}\, \text{M}$ | $1.33_{537}\times 10^{-2}\, \text{M}$ | $1.33_{521}\times 10^{-2}\, \text{M}$ | ||
| Change in Concentrations: | +x | -x | -x | ||
| Equilibrium Concentrations: | $4.29_{136}\times 10^{-3} + x$ | $1.33_{537}\times 10^{-2} – x $ | $1.33_{521}\times 10^{-2} – x $ |
$$K_C = = \frac{[SbCl_3][Cl_2]}{[SbCl_5]}\\
0.0249_{293} = \frac{[1.33_{537}\times 10^{-2} – x][1.33_{521}\times 10^{-2} – x]}{[4.29_{136}\times 10^{-3} + x]} \\
0 = x^2 – 0.0516_{351}x + 7.1_3193 \times 10^{-5} \\
x = 1.42028 \times 10^{-3}, 5.02148 \times 10^{-2}$$
Only the first value for $x$ above makes chemical sense:
$$[SbCl_5] = 4.29_{136}\times 10^{-3} + x = 4.29_{136}\times 10^{-3} + 1.42028 \times 10^{-3} \\
= 5.71_{944}\times 10^{-3}\, \text{M}\times \frac{299.0_5 \, \text{g}\, SbCl_5}{1\, \text {mol}}\times 3.00\, \text{L} = \mathbf{5.13\, \text{g}\, SbCl_5}\\
[SbCl_3] = 1.33_{537}\times 10^{-2} – x = 1.33_{537}\times 10^{-2} – 1.42028 \times 10^{-3} \\
= 0.0119_{2562}\, \text{M} \times \frac {228.1_5 \, \text{g}\, SbCl_3}{1\, \text {mol}}\times 3.00\, \text{L} = \mathbf{8.16\, \text{g}\, SbCl_3}\\
[Cl_2] = 1.33_{521}\times 10^{-2} – x = 1.33_{521}\times 10^{-2} – 1.42028 \times 10^{-3} \\
= 0.0119_{2402}\, \text{M} \times \frac {70.90 \, \text{g}\, Cl_2}{1\, \text {mol}}\times 3.00\, \text{L} = \mathbf{2.54\, \text{g}\, Cl_2}$$
Reality check 1: Decreasing the volume of this reaction at equilibrium should cause the reaction to shift left (towards the reactant) since there are fewer mol of gas on the reactant side of the equation. This matches our final concentrations.
Reality check 2: calculate K with the new concentrations: K = 0.0249 (agrees).
Consider the equilibrium
$$4NO_2(g)+6H_2O(g) \rightleftharpoons 4NH_3(g)+7O_2(g)$$(a) What is the expression for the equilibrium constant (Kc) of the reaction?
Solution
$$K_C = \frac{[NH_3]^4 [O_2]^7}{[NO_2]^4 [H_2 O]^6}$$
(b) How must the concentration of NH3 change to reach equilibrium if the reaction quotient is less than the equilibrium constant?
Solution
If Q < K, the reaction has “too few” products (or “too many” reactants). The reaction must proceed to the right (towards the products) to reach equilibrium. The concentration of NH3 will therefore increase.
(c) If the reaction were at equilibrium, how would an increase in the volume of the reaction vessel affect the pressure of NO2?
Solution
Initially (after the change but before the reaction comes to equilibrium) all pressures would decrease due to the incease in volume. In order to re-establish equilibrium, the reaction will proceed towards the side with more mol of gas (towards the products), so once equilibrium is re-established after the change, the pressure of NO2 will be lower than it was before the change.
(d) If the change in the pressure of NO2 is 0.28 bar as a mixture of the four gases reaches equilibrium, how much will the pressure of O2 change?
Solution
Changes in pressure are all related stoichiometrically; if ΔNO2 = “4x” then ΔO2 = “-7x”. Therefore, if the pressure of NO2 changes by 0.28 bar, the O2 will change by 0.49 bar in the opposite direction.
The binding of oxygen by hemoglobin (Hb), giving oxyhemoglobin (HbO2), is partially regulated by the concentration of H3O+ and dissolved CO2 in the blood. Although the equilibrium is complicated, it can be summarized as
$$HbO_2\,(aq)+H_3O^+\, (aq)+CO_2\, (aq) \rightleftharpoons (CO_2−Hb−H^+ )\, (aq) + O_2\,(aq)+H_2O\,(l)$$(a) Write the equilibrium constant expression $K_C$ for this reaction.
Solution
$$K_C = \frac{[CO_2−Hb−H^+ ][O_2]}{[HbO_2][H_3O^+][CO_2]}$$
(b) Explain why the production of lactic acid and CO2 in a muscle during exertion stimulates release of O2 from the oxyhemoglobin in the blood passing through the muscle.
Solution
Production of lactic acid (as you can infer from the name) will lower the pH of the blood, increasing the concentration of $H_3 O^+$. This is a reactant in the equilibrium expression above, so its addition will cause the reaction to shift towards the right (products), releasing O2 in the affected area.
Liquid N2O3 is dark blue at low temperatures, but the color fades and becomes greenish at higher temperatures as the compound decomposes to NO and NO2. At 25 °C, a value of KP = 1.91 has been established for this decomposition. If 0.236 moles of N2O3 are placed in a 1.52-L vessel at 25 °C, calculate the equilibrium partial pressures of N2O3(g), NO2(g), and NO(g). Assume all species are gaseous at this temperature.
Solution
$$N_2 O_3\, (g) \rightleftharpoons NO\, (g)\, + NO_2 (g)$$ Intial pressure of $N_2 O_3$: $$ PV = nRT \\ P = \frac{0.236\, \text{mol} \times 0.08314510 \frac{\text{L bar}}{\text{mol K}} \times 298\, \text{K}}{1.52\, \text{L}}\\ P = 3.84_{6992}\, \text{bar}\, N_2 O_3$$ The reaction container is initially prepared with only $N_2 O_3$ (the reactant) so the reaction must proceed to the right (towards the products) to reach equilibrium.
| $N_2 O_3\, (g)$ | $\rightleftharpoons $ | $NO\, (g) $ | + | $NO_2 \, (g)$ | |
| Initial Pressures: | $3.84_{6992}\, \text{bar}$ | 0 | 0 | ||
| Change in Pressures: | -x | +x | +x | ||
| Equilibrium Pressures: | 3.846992 – x | x | x |
A 1.00-L vessel at 400 °C contains the following equilibrium concentrations: N2, 1.00 M; H2, 0.50 M; and NH3, 0.25 M. How many moles of hydrogen must be removed from the vessel to increase the concentration of nitrogen to 1.1 M? The equilibrium reaction is $$N_2(g)+3\, H_2(g) \rightleftharpoons 2\, NH_3(g)$$
Solution
Determining the initial value for K: $$K_C = \frac{[NH_3]^2}{[N_2][H_2]^3}\\ K_C = \frac{[0.25]^2}{[1.00][0.50]^3}\\ K_C = 0.50$$ We can consider the amount of hydrogen gas removed from the container as $n$ M, and the change required to re-establish equilibrium as $x$ mol. Organizing the information given in an ICE table:
| $N_2\, (g)$ | + | $3\, H_2\, (g)$ | $\rightleftharpoons $ | $2\, NH_3\, (g) $ | |
| Initial Concentrations: | 1.00 M | (0.50 M – n) | 0.25 M | ||
| Change in Concentrations: | +x | +3x | -2x | ||
| Equilibrium Concentrations: | 1.00 + x = 1.1 M |
(0.50 – n) + 3x | 0.25 – 2x |
We can see by looking at the nitrogen concentration that $x = 0.1\, \text{M}$. Substituting this value for $x$ into the other concentrations, and into the expression for $K$: $$K_C = \frac{[NH_3]^2}{[N_2][H_2]^3}\\ 0.50 = \frac{[0.25 – 2(0.1)]^2}{[1.1][(0.50 – n) + 3(0.1)]^3}\\ 0.50 = \frac{[0.0025]}{[1.1][0.80-n]^3}\\ [0.80-n]^3 = 0.0045_{4545}\\ 0.80-n = 0.16_{565}\\ n = 0.63_{434}$$ You would have to remove 0.63 M of H2 in order to reach the target equilibrium state.