The Periodic Table
The modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 2.26). Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. The elements are arranged in seven horizontal rows, called periods or series, and 18 vertical columns, called groups. Groups are labelled at the top of each column. IUPAC recommends that the numbers 1 through 18 be used, and thus this labelling is more common. For the table to fit on a single page, parts of two of the rows, a total of 14 columns, are usually written below the main body of the table.
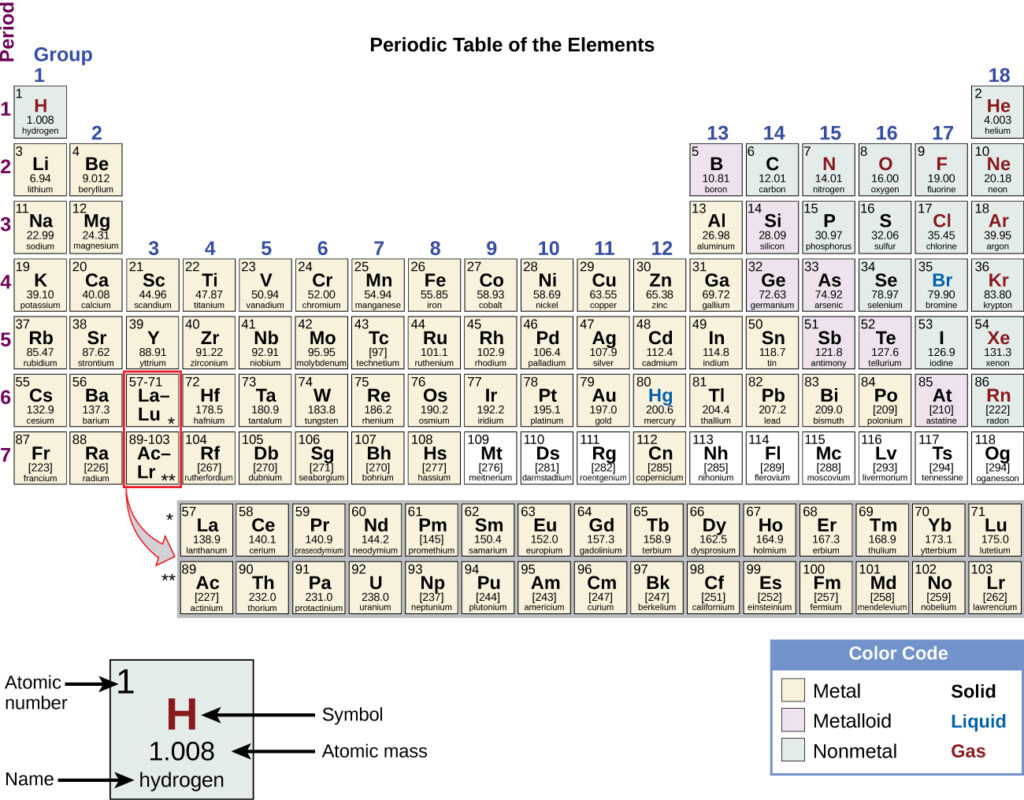
We can sort the elements into large classes with common properties: metals (elements that are shiny, malleable, good conductors of heat and electricity—shaded yellow); nonmetals (elements that appear dull, poor conductors of heat and electricity—shaded green); and metalloids (elements that conduct heat and electricity moderately well, and possess some properties of metals and some properties of nonmetals—shaded purple).

The elements can also be classified into the main-group elements (or representative elements) in the columns labeled 1, 2, and 13–18; the transition metals in the columns labeled 3–121; and inner transition metals in the two rows at the bottom of the table (the top-row elements are called lanthanides and the bottom-row elements are actinides; Figure 2.27). The elements can be subdivided further by more specific properties, such as the composition of the compounds they form. For example, the elements in group 1 (the first column) (except hydrogen) are known as alkali metals, and they all have similar chemical properties. The elements in group 2 (the second column) are called alkaline earth metals, with similar properties among members of that group. Other groups with specific names commonly used are the pnictogens, (group 15), chalcogens (group 16), halogens (group 17), and the noble gases (group 18). Hydrogen is a unique, nonmetallic element with properties similar to both group 1 and group 17 elements. For that reason, hydrogen may be shown at the top of both groups, or by itself.
Click on this link for an interactive periodic table, which you can use to explore the properties of the elements (includes podcasts and videos of each element). You may also want to try this one that shows photos of all the elements.
The atomic masses on the periodic table reflect the weighted average mass of the element in its natural state – e.g. the average mass of a natural sample of carbon is (approximately) 12.01 grams per mole. This is based on the mass of each isotope (see the next page on atomic structure) and their relative abundance on earth. Review calculating weighted averages here.
In studying the periodic table, you might have noticed something about the atomic masses of some of the elements. Element 43 (technetium), element 61 (promethium), and most of the elements with atomic number 84 (polonium) and higher have their atomic mass given in square brackets. This is done for elements that consist entirely of unstable, radioactive isotopes. An average atomic weight cannot be determined for these elements because their radioisotopes may vary significantly in relative abundance, depending on the source – or may not even exist in nature. The number in square brackets is the atomic mass number (an approximate atomic mass) of the most stable isotope of that element.
Footnotes
- 1 Per the IUPAC definition, group 12 elements are not transition metals, though they are often referred to as such.