Key Concepts and Summary
Integrated rate laws are determined by integration of the corresponding differential rate laws. Rate constants for those rate laws are determined from measurements of concentration at various times during a reaction.
The half-life of a reaction is the time required to decrease the amount of a given reactant by one-half. The half-life of a zero-order reaction decreases as the initial concentration of the reactant in the reaction decreases. The half-life of a first-order reaction is independent of concentration, and the half-life of a second-order reaction decreases as the concentration increases.
[table id=38 /]
Key Equations
- integrated rate law for zero-order reactions: $[A]_t=-kt+[A]_0$,
- half-life for a zero-order reaction $t_{1/2}=\frac{[A]_0}{2k}$
- integrated rate law for first-order reactions: $ln[A]_t=-kt+ln[A]_0$,
- half-life for a first-order reaction $t_{1/2}=\frac{0.693}{k}$
- integrated rate law for second-order reactions: $\frac{1}{[A]_t}=kt+\frac{1}{[A]_0}$,
- half-life for a second-order reaction $t_{1/2}=\frac{1}{[A]_0k}$
Practice Problems: Integrated Rate Laws
Describe how graphical methods can be used to determine the order of a reaction and its rate constant from a series of data that includes the concentration of A at varying times.
Solution:
To determine the order of reaction (for a reactant “A”), you would make (up to) three plots:
- [A] vs t
- ln[A] vs t
- $\frac{1}{[A]}$ vs t
- 0th order: $[A]_t = -kt + [A]_0$ ([A] vs t)
- 1st order: $ln[A]_t = -kt + ln[A]_0$ (ln[A] vs t)
- 2nd order: $\frac{1}{[A]_t} = kt + \frac{1}{[A]_0}$ ($\frac{1}{[A]}$ vs t)
Use the data provided to graphically determine the order and rate constant of the following reaction: $$SO_2Cl_2⟶SO_2+Cl_2$$
| Time (s) | $0.00$ | $5.00×10^3$ | $1.00×10^4$ | $1.50×10^4$ | $2.50×10^4$ | $3.00×10^4$ | $4.00×10^4$ |
| [SO2Cl2] (M) | 0.100 | 0.0896 | 0.0802 | 0.0719 | 0.0577 | 0.0517 | 0.0415 |
Solution:
First, we should make the “test plots” for the 3 available reaction orders:
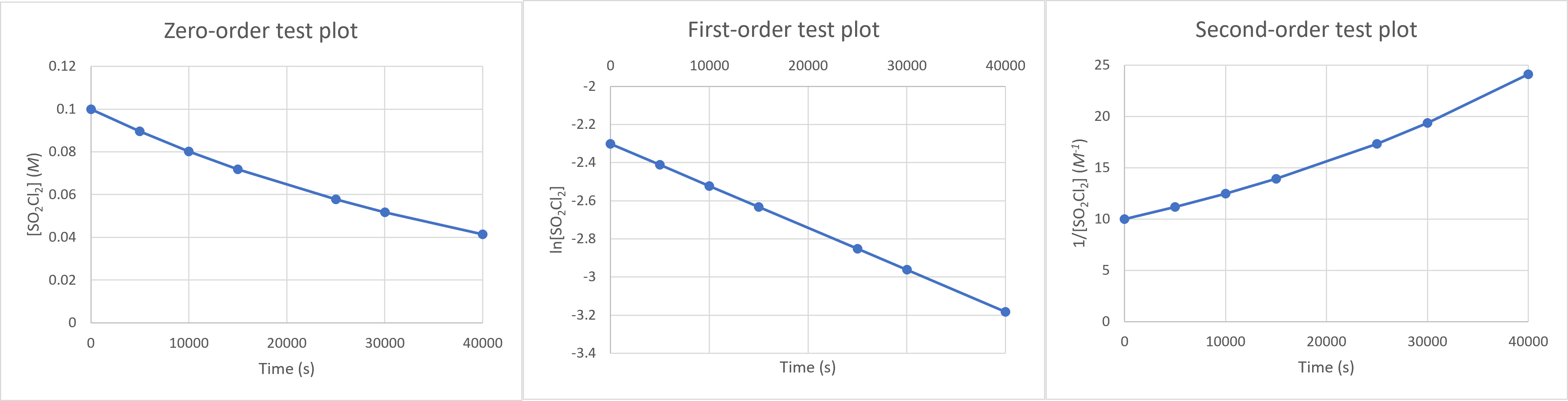 The ln[SO2Cl2] vs time plot is linear, which corresponds to the first-order integrated rate law. The reaction is first-order in SO2Cl2.
The ln[SO2Cl2] vs time plot is linear, which corresponds to the first-order integrated rate law. The reaction is first-order in SO2Cl2.
Finding the slope of this plot (using any 2 points on the line) will give $-k$, according to the linearized first-order rate law $ln[A]_t = -kt + ln[A]_0$. The rate constant is $\mathbf{k = 2.20\times 10^{−5} s^{−1}}$.
Pure ozone decomposes slowly to oxygen, $2\, O_3(g)\rightarrow 3\, O_2(g)$. Use the data provided in a graphical method and determine the order and rate constant of the reaction.
| Time (h) | 0 | 2.0 × 103 | 7.6 × 103 | 1.00 × 104 | 1.23 × 104 | 1.43 × 104 | 1.70 × 104 |
| [O3] (M) | 1.00 × 10−5 | 4.98 × 10−6 | 2.07×10−6 | 1.66 × 10−6 | 1.39 × 10−6 | 1.22 × 10−6 | 1.05 × 10−6 |
Solution:
First, we should make the “test plots” for the 3 available reaction orders: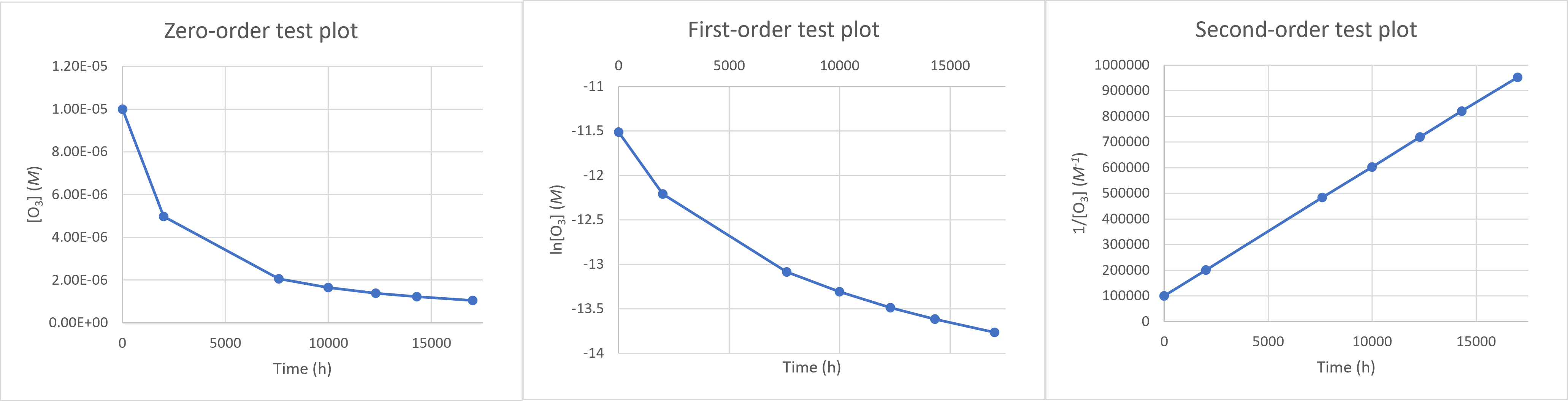 The 1/[O3] vs time plot is linear, which corresponds to the second-order integrated rate law. The reaction is second-order in O3.
The 1/[O3] vs time plot is linear, which corresponds to the second-order integrated rate law. The reaction is second-order in O3.Finding the slope of this plot (using any 2 points on the line) will give $k$, according to the linearized second-order rate law $\frac{1}{[A]_t} = -kt + \frac{1}{[A]_0}$. The rate constant is $\mathbf{k = 50.2\, L\, mol^{-1}h^{-1}}$.
Think about: Why does the first time point “0” not change the number of significant figures in the answer? How will you determine the correct number of significant figures in your value for k?
From the data given below, use a graphical method to determine the order and rate constant of the following reaction: $2\,X \rightarrow Y+Z$.
| Time (s) | 5.0 | 10.0 | 15.0 | 20.0 | 25.0 | 30.0 | 35.0 | 40.0 |
| [X] (M) | 0.0990 | 0.0497 | 0.0332 | 0.0249 | 0.0200 | 0.0166 | 0.0143 | 0.0125 |
Solution:
First, we should make the “test plots” for the 3 available reaction orders: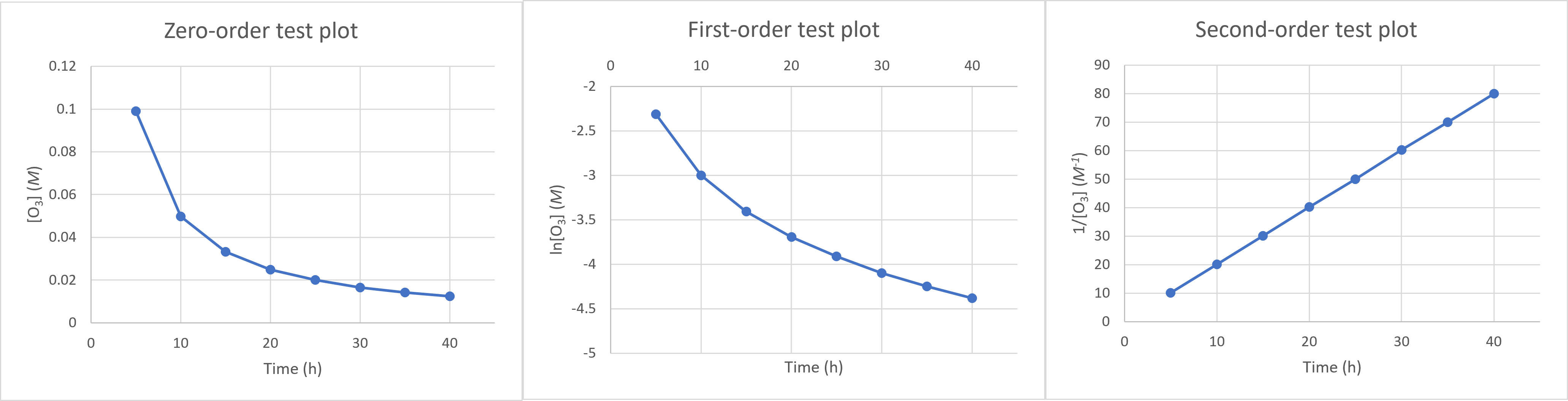 The 1/[X] vs time plot is linear, which corresponds to the second-order integrated rate law. The reaction is second-order in X.
The 1/[X] vs time plot is linear, which corresponds to the second-order integrated rate law. The reaction is second-order in X.Finding the slope of this plot (using any 2 points on the line) will give $k$, according to the linearized second-order rate law $\frac{1}{[A]_t} = -kt + \frac{1}{[A]_0}$. The rate constant is $\mathbf{k = 2.00\: L\, mol^{-1}s^{-1}}$.
What is the half-life for the first-order decay of phosphorus-32? $(_{15}^{32}P ⟶ _{16}^{32}S +e−)$ The rate constant for the decay is $4.85\times 10^{−2}\:\text{day}^{-1}$.
Solution:
From the units of the rate constant, we know this reaction follows first-order kinetics.
Using the formula for first-order half life, and the rate constant provided:
$$t_{\frac{1}{2}} = \frac{ln\, 2}{k} \\
t_{\frac{1}{2}} = \frac{ln\, 2}{4.85\times 10^{−2}\:\text{day}^{-1}} \\
t_{\frac{1}{2}} = 14.2_{917}\:\text{days}\\
\mathbf{t_{\frac{1}{2}} = 14.3\:\text{days}} $$
What is the half-life for the first-order decay of carbon-14? $(_6^{14}C ⟶ _7^{14}N +e−)$ The rate constant for the decay is $1.21\times 10^{-4}\: \text{year}^{-1}$.
Solution:
From the units of the rate constant (and the question text!), we know this reaction follows first-order kinetics.
Using the formula for first-order half life, and the rate constant provided:
$$t_{\frac{1}{2}} = \frac{ln\, 2}{k} \\
t_{\frac{1}{2}} = \frac{ln\, 2}{1.21\times 10^{-4}\: \text{year}^{-1}} \\
t_{\frac{1}{2}} = 5.72_{849}\times 10^{3}\:\text{years}\\
\mathbf{t_{\frac{1}{2}} = 5.73\times 10^{3}\:\text{years}} $$
What is the half-life for the decomposition of NOCl when the concentration of NOCl is 0.15 M? The rate constant for this second-order reaction is $8.0 \times 10^{-8}\: L\, mol^{-1}s^{-1}$.
Solution:
From the units of the rate constant (and the question text!), we know this reaction follows second-order kinetics.
Using the formula for second-order half life, and the data provided:
$$t_{\frac{1}{2}} = \frac{1}{k\, [A]_0}\\
t_{\frac{1}{2}} = \frac{1}{0.15\, M \cdot 8.0 \times 10^{-8} M^{-1}s^{-1}} \\
t_{\frac{1}{2}} = 83_{333333.33}\:\text{s}\\
\mathbf{t_{\frac{1}{2}} = 8.3\times 10^{7}\:\text{s}} \quad \text{OR}\; 2.3\times 10^4\: \text{hours} \quad \text{OR}\: 9.6 \times 10^2 \text{days} $$
What is the half-life for the decomposition of O3 when the initial concentration of O3 is $2.35 \times 10^{-6}\: M$? The rate constant for this reaction is $50.4\: L\, mol^{-1}\, h^{-1}$.
Solution:
From the units of the rate constant, we know this reaction follows second-order kinetics.
Using the formula for second-order half life, and the data provided:
$$t_{\frac{1}{2}} = \frac{1}{k\, [A]_0}\\
t_{\frac{1}{2}} = \frac{1}{2.35 \times 10^{-6}\, M \cdot 50.4\: M^{-1}\, h^{-1}} \\
t_{\frac{1}{2}} = 844_{3.094}\:\text{h}\\
\mathbf{t_{\frac{1}{2}} = 8.44\times 10^{3}\:\text{h}} \quad \text{OR} \: 352\, \text{days} $$
The reaction of compound A to give compounds C and D was found to be second-order in A. The rate constant for the reaction was determined to be $2.42\: L\, mol^{-1} s^{-1}$. If the initial concentration is 0.500 mol/L, what is the value of t1/2?
Solution:
Using the formula for second-order half life, and the data provided: $$t_{\frac{1}{2}} = \frac{1}{k\, [A]_0}\\ t_{\frac{1}{2}} = \frac{1}{2.42\: M^{-1} s^{-1} \cdot 0.500 M} \\ t_{\frac{1}{2}} = 0.826_{446}\:\text{s}\\ \mathbf{t_{\frac{1}{2}} = 0.826\:\text{s}}$$
The half-life of a reaction of compound A to give compounds D and E is 8.50 min when the initial concentration of A is 0.150 M. How long will it take for the concentration to drop to 0.0300 M if the reaction is: (a) first order with respect to A
Solution:
In order to solve for time elapsed in a reaction, we will use the integrated rate law. For a first order reaction, this is:
$$ln[A]_t = -kt + ln[A]_0 $$
From the question text, we know $[A]_t$ (0.0300 M) and $[A]_0$ (0.150 M), but we do not know $k$, which is also needed in order to solve for $t$ in this equation.
We also know the half-life of the reaction. The formula for a first-order half-life is:
$$t_{\frac{1}{2}} = \frac{ln(2)}{k}$$
We can use the half-life to solve for $k$:
$$8.50\, \text{min} = \frac{ln(2)}{k}$\\
k = 0.0815_{467} \text{min}^{-1}$$
Combining this $k$ with the concentration data provided in the first-order integrated rate law:
$$ln[A]_t = -kt + ln[A]_0 \\
ln(0.0300\: M) = -(0.0815_{467} \text{min}^{-1})t + ln(0.150\: M) \\
{t = 19.7_{364} \text{min}} \\
\mathbf{t = 19.7 \text{min}} $$
Solution:
Following the same logic as in part (a) above: $$ t_{\frac{1}{2}} = \frac{1}{k[A]_0} \\ 8.50\: \text{min} = \frac{1}{k(0.150\: M)} \\ k = 0.784_{314}\: \text{min}^{-1}$$ $$\frac{1}{[A]_t} = kt + \frac{1}{[A]_0} \\ \frac{1}{0.0300\: M} = (0.784_{314}\: \text{min}^{-1})t + \frac{1}{0.150\: M} \\ t = 33.9_{999}\: \text{min} \\ \mathbf{t = 34.0\: \text{min}} $$
Some bacteria are resistant to the antibiotic penicillin because they produce penicillinase, an enzyme with a molecular weight of 3×104 g/mol that converts penicillin into inactive molecules. Although the kinetics of enzyme-catalyzed reactions can be complex, at low concentrations this reaction can be described by a rate law that is first order in the catalyst (penicillinase) and that also involves the concentration of penicillin. From the following data: 1.0 L of a solution containing 0.15 µg (0.15×10−6 g) of penicillinase, determine the order of the reaction with respect to penicillin and the value of the rate constant.
| [Penicillin] (M) | Rate (mol L−1 min−1) | |
|---|---|---|
| Run 1 | 2.0 × 10−6 | 1.0 × 10−10 |
| Run 2 | 3.0 × 10−6 | 1.5 × 10−10 |
| Run 3 | 4.0 × 10−6 | 2.0 × 10−10 |
Solution
We know from the question text that the rate law is first-order in penicillinase, and also depends on the penicillin concentration. A preliminary version of the rate law can be written as: $$ \text{rate} = k[\text{penicillinase}][\text{penicillin}]^m $$ Since the concentration of penicillinase is constant, we can solve for the order of penicillin in one of two ways:
Method 1: Pseudo-order kinetics
Since the concentration of penicillinase is constant, we can “roll it into” the rate constant to make a pseudo-rate constant $k’$. This makes the rate law:
$$ \text{rate} = k’\,[\text{penicillin}]^m $$
Then, since we can see that the rate will now only depend on one changing concentration, we can directly compare two of the experiments to determine the order.
Using Runs 1 and 3: the concentration of penicillin has doubled. The rate has also doubled. This means the reaction is first order in penicillin.
Method 2: Ratios
We can also use the method of ratios as described in the Method of Initial Rates section.
$$\frac{\text{rate 1}}{\text{rate 2}} = \frac{[\text{penicillinase}]_1[\text{penicillin}]_1 ^m}{[\text{penicillinase}]_2[\text{penicillin}]_2 ^m} \\
\frac{1.0\times 10^{-10}}{1.5 \times 10^{-10}} = \frac{[5\times 10^{-12} M]^* [2.0 \times 10^{-6} M]^m }{[5\times 10^{-12} M][3.0 \times 10^{-6} M]^m } \\
0.66_{66667} = 1 \times \frac{[2.0 \times 10^{-6} M]^m }{[3.0 \times 10^{-6} M]^m } \\
ln (0.66_{66667}) = m\cdot ln\left( \frac{[2.0 \times 10^{-6} M]}{[3.0 \times 10^{-6} M]} \right) \\
1 = m $$
* This concentration is determined from the mass concentration (μg/L) and molecular mass given in the question.
The reaction is first order in penicillin.
Finding $k$
To find the rate constant $k$, we return to the full rate law (no pseudo-rate laws, if used) and substitute in data from any of the experiments to solve for $k$:
$$ \text{rate} = k[\text{penicillinase}][\text{penicillin}] \\
1.0 \times 10^{-10} \frac{M}{\text{min}} = k[5\times 10^{-12}\, M] [2.0 \times 10^{-6}\, M] \\
k = 1.0 \times 10^7 \, \frac{1}{M\cdot \text{min}}
$$
Both technetium-99 and thallium-201 are used to image heart muscle in patients with suspected heart problems. The half-lives are 6.0 h and 73 h, respectively. What percent of the original isotope would remain for each of these species after exactly 2 days?
Hint: You may want to revisit the last time the half life of radioactive elements was discussed.Solution
From the link above, all radioactive decay follows first-order kinetics. This means the half-life of each element is constant, and independent of the amount of starting material.
For $\mathbf{^{99}Tc}$:
There are two ways to approach this problem.
Method 1: Number of half livesSince 6.0h divides into 2 days (48 hours) evenly, we can simply use the number of half lives to determine the amount that will remain. After 1 hafl life, 50% of the initial amount remains. After 2 half lives, 50% of that remains (0.50 * 0.50) = 0.25 or 25%. You can keep going this way, or use the general form of (0.5)n where n is the number of half-lives. To get to 48h, this is 8.0 half-lives, so $$0.5^{8.0} = 0.00390625$$ or 0.39% will remain. (Two significant figures from the 6.0 h – the “50%” or “0.5” in the definition of half-life is exact).
Method 2: Integrated rate law
More generally, we can always use the integrated rate law for a reaction to determine concentration at a given time (assuming we have sufficient information). The integrated rate law for a first-order reaction is:
$$ ln[A]_t = -kt + ln[A]_0 $$
Initially, we do not know the value for k, but we can find it from the half life (again for a first order reaction) and the information provided:
$$ t_{\frac{1}{2}} = \frac{ln(2)}{k} \\
6.0\, \text{h} = \frac{ln(2)}{k} \\
k = 0.11_{55245} \, \text{h}^{-1} $$
Now, we can use this $ k $ value in the integrated rate law. Since we are looking for a percentage remaining, we can use “1” or “100” as our initial concentration to make the calculation easy, since no initial concentration is given.
$$ ln[A]_t = -kt + ln[A]_0 \\
ln[A]_t = – \left(0.11_{55245}\, \text{h}^{-1} \right) \left( 48\, \text{h} \right) + ln[100] \\
ln[A]_t = -0.94_{0007258} \\
[A]_t = 0.39_{0625} \\
\text{percent remaining} = \frac{0.39}{100}\times 100\% \\
\mathbf{\text{percent remaining} = 0.39\% }$$
For $\mathbf{^{201}Tl}$:
Method 1:
The 2 days (48h) half-life is 0.657554 times the 73h time frame we are calculating for. This gives $0.5^{0.65_{7534}}$ or 63% remaining (63.3960%).
Method 2:
From the first order half-life equation, we find $k = 9.4_{952}\times 10^{-3}\, \text{h}^{-1}$, and from the integrated rate law, 63% remains (63.3960%).
There are two molecules with the formula C3H6. One isomer, n-propene $(CH_3 CH=CH_2)$ is is the monomer used to make polypropylene, a common polymer that is used to make indoor-outdoor carpets and plastic bottles. Near Redwater, Alberta is a large polypropylene manufacturing site.
The other isomer, cyclopropane, is used as an anesthetic:
From the units of the rate constant, we can tell that this reaction is first-order in cyclopropane. Using the first-order half-life expression:
$$ t_\frac{1}{2} = \frac{ln(2)}{k} \\
t_\frac{1}{2} = \frac{ln(2)}{5.95\times 10^{-4}\, \text{s}^{-1}} \\
t_\frac{1}{2} = 1164.9532\, \text{s} \\
\mathbf{t_\frac{1}{2} = 1.16 \times 10^3\, \text{s}} $$
 When heated to 499 °C, cyclopropane rearranges (isomerizes) and forms propene with a rate constant of $5.95\times 10^{-4}\, \text{s}^{-1}$.
When heated to 499 °C, cyclopropane rearranges (isomerizes) and forms propene with a rate constant of $5.95\times 10^{-4}\, \text{s}^{-1}$.
(a) What is the half-life of this reaction?
Solution
Solution
Using the first-order integrated rate law, and the $k$ calculated above: $$ ln[A]_t = -kt + ln[A]_0 \\ ln[A]_t = – \left(5.95\times 10^{-4} \text{s}^{-1} \right) \times 0.75\, \text{h} \frac{3600 \, \text{s}}{1 \, \text{h}} + ln[100 \, \text M] \\ ln [A]_t = 2.99_{8670} \\ [A]_t = 20._{058845}\, \text{M} \\ \frac{20._{058845}\, \text{M}}{100 \, \text{M}} \times 100\% = 20._{058845}\% \\ \mathbf{\text{Amount remaining}= 20.\% \, \text{of the initial value}} $$ (Since we are not given an initial amount, but we are looking only for a relative amount remaining (i.e. a %), we can use an arbitrary amount for [A]0. “1” or “100” is convenient if we are looking for a fraction!)
Fluorine-18 is a radioactive isotope that decays by positron emission to form oxygen-18 with a half-life of 109.7 min. (A positron is a particle with the mass of an electron and a single unit of positive charge; the equation is $_9^{18}F ⟶ _8^{18}O +_{+1}^0e$ Physicians use 18F to study the brain by injecting a quantity of fluoro-substituted glucose into the blood of a patient. The glucose accumulates in the regions where the brain is active and needs nourishment.
(a) What is the rate constant for the decomposition of fluorine-18?
Solution
All radioactive decay has first-order kinetics. We can use the half-life to find $k$: $$ t_\frac{1}{2} = \frac{ln(2)}{k} \\ 109.7 \, \text{min} = \frac{ln(2)}{k} \\ k = 6.318_{57047}\times 10^{-3}\, \text{min}^{-1} \\ \mathbf{k = 6.319 \times 10^{-3}\, \text{min}^{-1}} $$
(b) If a sample of glucose containing radioactive fluorine-18 is injected into the blood, what percent of the initial $^{18}F$ isotope will remain after 5.59 h?
Solution
Using the first-order integrated rate law, and the $k$ calculated above: $$ ln[A]_t = -kt + ln[A]_0 \\ ln[A]_t = – \left( 6.318_{57047}\times 10^{-3}\, \text{min}^{-1} \right)\left( 5.59\, \text{h} \frac{60\, \text{min}}{1\, \text{h}} \right) + ln[100\, \text{M}] \\ ln[A]_t = 2.4859_{2165} \\ [A]_t = 12.0_{12186}\, \text{M} \\ \frac{12.0_{12186}\, \text{M}}{100\, \text{M}} \times 100\% = 12.0_{12186}\% \\ \mathbf{\text{Amount remaining = 12.0%}}$$
(c) How long does it take for 99.99% of the 18F to decay?
Solution
99.99% remaining means that 0.01% remains (or if we started with 100.00 M, 0.01 M would remain). $$ ln[A]_t = -kt + ln[A]_0 \\ ln[0.01\, \text{M}] = – \left( 6.318_{57047}\times 10^{-3}\, \text{min}^{-1} \right)t + ln[100\, \text{M}] \\ t = 1._{457662}\times 10^{3}\, \text{min} \\ \mathbf{t = 1\times 10^{3}\, \text{min}}\text{ OR } 20\, \text{h OR }1\, \text{day} $$
Suppose that the half-life of a steroid taken by an athlete is 42 days. Assuming that the steroid biodegrades via a first-order process, how long would it take for $\frac{1}{64}$ of the initial dose to remain in the athlete’s body?
Solution
We do not have a $k$ for this reaction, so we cannot use the integrated rate law directly. However $\frac{1}{64}$ is an even number of half-lives: $$\frac{1}{64} = \left( \frac{1}{2} \right)^6$$ Therefore, it will take 6 half-lives, or 252 days to reach this point.
In 2012, the skeleton of King Richard III was found under a parking lot in Leicester, England. If tissue samples from the skeleton at that time contained about 93.79% of the carbon-14 expected in living tissue, what year did King Richard III die? The half-life for the decay of carbon-14 is 5730 years.
Solution
Radiocarbon dating is based on the radioactive decay of $^{14}C$, therefore it is a first-order process.
First, we find $k$ from the half-life:
$$t_\frac{1}{2} = \frac{ln(2)}{k} \\
5730\, \text{years} = \frac{ln(2)}{k} \\
k = 1.209_{6809}\times 10^{-4}\, \text{years}^{-1}$$
Then bring this into the first-order integrated rate law:
(This time, we have 93.79% remaining, not 93% consumed)
$$ ln[A]_t = -kt + ln[A]_0 \\
ln[93.79] = -\left( 1.209_{6809}\times 10^{-4}\, \text{years}^{-1} \right)t + ln[100] \\
t = 529.9_{905}\text{years} $$
So the sample is about 530 years old at the time of sampling, meaning the skeleton would have died in about 1482 CE. Richard III died in 1485, so this data would be consistent with the skeleton belonging to Richard III, given that radiocarbon dating is precise to with about ±30 years. The actual results do confirm the skeleton’s identity as Richard III (and confirmed by genetic analysis).
Nitroglycerine is an extremely sensitive explosive. In a series of carefully controlled experiments, samples of the explosive were heated to 160 °C and their first-order decomposition studied. Determine the average rate constants for each experiment using the following data:
| Initial [C3H5N3O9] (M) | 4.88 | 3.52 | 2.29 | 1.81 | 5.33 | 4.05 | 2.95 | 1.72 |
| t (s) | 300. | 300. | 300. | 300. | 180. | 180. | 180. | 180. |
| % Decomposed | 52.0 | 52.9 | 53.2 | 53.9 | 34.6 | 35.9 | 36.0 | 35.4 |
Solution
For each experiment, you can find $k$ from the first order rate law expression. For the first data set:
$$ ln[A]_t = -kt + ln[A]_0 \\
ln\left( 48.0\% \times 4.88\, \text{M} \right) = -k(300\, \text{s}) + ln(4.88\, \text{M}) \\
k = 2.44_{65639} \times 10^{-3}\, \text{s}^{-1} \\
k = 2.45 \times 10^{-3}\, \text{s}^{-1}$$
Note that the table gives you % Decomposed, while the integrated rate law requires the amount remaining in the reaction (so 52% decomposed → 48% remaining).
Results for the rest of the data:
[A]0 (M)
k × 10-3 (s−1)
4.88
2.45
3.52
2.51
2.29
2.53
1.81
2.58
5.33
2.36
4.05
2.47
2.95
2.48
1.72
2.43
For the past 10 years, the unsaturated hydrocarbon 1,3-butadiene $(CH_2 = CH-CH=CH_2 )$ has ranked 38th among the top 50 most used industrial chemicals. It is used primarily for the manufacture of synthetic rubber. A cyclic isomer also exists (cyclobutene):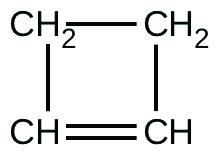
The isomerization of cyclobutene to butadiene is first-order and the rate constant has been measured as 2.0×10−4 s−1 at 150 °C in a 0.53 L flask. Determine the partial pressure of cyclobutene and its concentration (in mol/L) after 30.0 minutes if an isomerization reaction is carried out at 150 °C with an initial pressure of 7.5 kPa.