Second-Order Reactions
The equations that relate the concentrations of reactants and the rate constant of second-order reactions are fairly complicated. We will limit ourselves to the simplest second-order reactions, namely, those with rates that are dependent upon just one reactant’s concentration and described by the differential rate law: $$rate=k[A]^2 \label{eq1}\tag{1}$$
For these second-order reactions, the integrated rate law is: $$\frac{1}{[A]_t}=kt+\frac{1}{[A]_0} \label{eq2}\tag{2}$$
where the terms in the equation have their usual meanings as defined when discussing rate laws earlier.
The Integrated Rate Law for a Second-Order Reaction
The reaction of butadiene gas (C4H6) with itself produces C8H12 gas as follows: $$2\;C_4 H_6 (g)\rightarrow C_8 H_{12} (g)$$
The reaction is second order with a rate constant equal to $ 5.76 \times 10^{-2}\; \frac{L}{mol\cdot min} $ (or $ 5.76 \times 10^{-2}\; \frac{1}{M\cdot min} $) under certain conditions. If the initial concentration of butadiene is 0.200 M, what is the concentration remaining after 10.0 min?
Solution:
We use the integrated form of the rate law to answer questions regarding time. For a second-order reaction, this is Equation $\ref{eq2}$ above.
We know three variables in this equation: [A]0 = 0.200 mol/L, $k= 5.76 \times 10^{-2}\; \frac{1}{M\cdot min} $ , and t = 10.0 min. Therefore, we can solve for the remaining variable, [A]t:
$$\frac{1}{[A]_t}=kt+\frac{1}{[A]_0} \\
\frac{1}{[A]_t}=( 5.76 \times 10^{-2}\; \frac{1}{M\cdot min} )(10\;min)+\frac{1}{0.200\;M}\\
\frac{1}{[A]_t}=(5.76 \times 10^{−1}\; \frac{1}{M})+5.00 \frac{1}{M} \\
\frac{1}{[A]_t}=5.58 \frac{1}{M} \\
[A]_t =1.79\times 10^{−1}\;M$$
Therefore 0.179 mol/L of butadiene remain at the end of 10.0 min, compared to the 0.200 mol/L that was originally present.
Check Your Learning
If the initial concentration of butadiene is 0.0200 M, what is the concentration remaining after 20.0 min? (Following the reaction described in the example above).
Answer:
Using Equation $\ref{eq2}$ and the data provided (k is given in the example above): $$\frac{1}{[A]_t}=kt+\frac{1}{[A]_0} \\ \frac{1}{[A]_t}=\left( 5.76 \times 10^{-2}\; \frac{L}{mol\cdot min} \right)\, (20.0\: \text{min}) + \frac{1}{0.0200\: M} \\ \frac{1}{[A]_t} = 51.1_{52}\: M^{-1} \\ [A]_t = 0.0195_{495}\: M \\ [A]_t = 0.0195\: M $$
The integrated rate law for our second-order reactions has the form of the equation of a straight line: $$\frac{1}{[A]_t}=kt+\frac{1}{[A]_0}\\y=mx+b$$
A plot of $\frac{1}{[A]_t}$ versus t for a second-order reaction is a straight line with a slope of k and an intercept of $\frac{1}{[A]_0}$. If the plot is not a straight line, then the reaction is not second order.
Determination of Reaction Order by Graphing
The data below are for the same reaction described in the previous example. Test these data to confirm that this dimerization reaction is second-order.
Solution
| Trial | Time (s) | [C4H6] (M) |
|---|---|---|
| 1 | 0 | 1.00 × 10−2 |
| 2 | 1600 | 5.04 × 10−3 |
| 3 | 3200 | 3.37 × 10−3 |
| 4 | 4800 | 2.53 × 10−3 |
| 5 | 6200 | 2.08 × 10−3 |
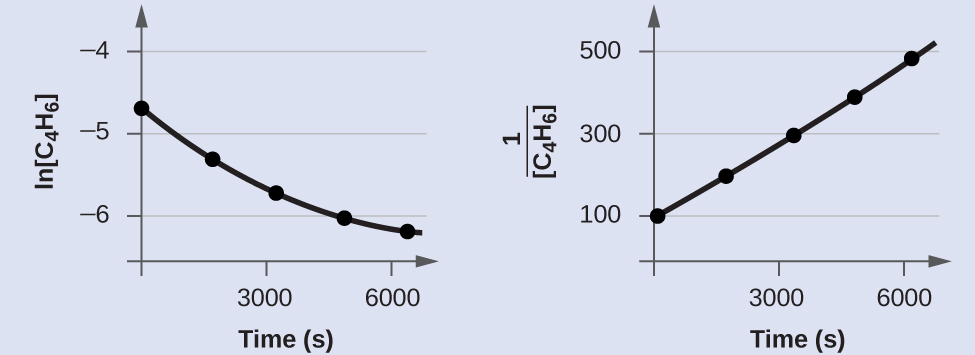
In order to distinguish a first-order reaction from a second-order reaction, we plot ln[C4H6] versus t and compare it with a plot of $\frac{1}{[C_4 H_ 6]}$ versus t. The values needed for these plots follow.
| Time (s) | $\frac{1}{[C _4 H_6]} $ (M−1) | ln[C4H6] |
|---|---|---|
| 0 | 100 | −4.605 |
| 1600 | 198 | −5.289 |
| 3200 | 296 | −5.692 |
| 4800 | 395 | −5.978 |
| 6200 | 481 | −6.175 |
The plots are shown below. As you can see, the plot of ln[C4H6] versus t is not linear, therefore the reaction is not first order. The plot of $\frac{1}{[C_4 H_6 ]}$ versus t is linear, indicating that the reaction is second order.
Check Your Learning
Does the following data describe a reaction with second-order kinetics?
Yes, this reaction is second-order in [A]. The plot of $\frac{1}{[A]}$ vs $t$ is linear:
Trial Time (s) [A] (M) 1 5 0.952 2 10 0.625 3 15 0.465 4 20 0.370 5 25 0.308 6 35 0.230 Answer:
