Reaction Rates Defined
A rate is a measure of how some property varies with time. You are probably familiar with some reates already: Speed is a rate that shows the distance an object travels in a set time. Wage is a rate that shows the money a person earns for working for a set time. In a similar way, the rate of a chemical reaction measures how much reactant is used or how much product is made in a set time.
The rate of reaction is the change in the amount of reactant or product per unit time. To find the reaction rate, you can measure how a measurable property (one that relates to the amount of a reactant or product) changes over time. For reactions that use or make gases, you can find the rate by measuring changes in partial pressure. For example, in the reaction $$2\:NO\:(g)\;+O_2\:(g)\;\rightarrow\;2\:NO_2\:(g)$$ the rate of reaction could be measured by monitoring the partial pressure of oxygen gas ($P_{O_2}$): $$\text{rate of reaction} \;=\; \frac{\text{change in pressure of oxygen}}{\text{change in time}}\\ =\;\frac{\Delta P_{O_2}}{\Delta t}$$
Remember that the symbol $\Delta$ indicates “change in” – and specifically calculated by taking $$\text{final value}\; -\; \text{initial value}$$
For reactants and products in solution, we normally use molar concentrations ($M$ or $\frac{mol}{L}$) when expressing reaction rates. For example, the concentration of hydrogen peroxide, H2O2, in an aqueous solution changes slowly over time as it decomposes according to the equation: $$2\; H_{2}O_{2}\;(aq) \rightarrow 2\;H_2 O \;(l)+ O_2\;(g)$$
The rate at which the hydrogen peroxide decomposes can be expressed in terms of the rate of change of its concentration: $$ rate\; of\; decomposition\; of\; H_2 O_2\; = \frac{change\; in\; concentration\; of\;reactant}{time\; interval} \\
=\frac{[H_2 O_2]_{t_2} – [H_2 O_2 ]_{t_1}}{t_2-t_1} \\
= \:- \frac{\Delta [H_2 O_2 ]}{\Delta t} $$
Types of Reaction Rates
Depending on what chemical species (reactant or product) is measured, and over what time, we can calculate different types of reaction rate.
Rate of Appearance
In a chemical reaction, the products are being created and their concentration (or partial pressure) will increase over time. The rate of appearance of a product (still $\frac{\Delta [\text{product}]}{\Delta [\text{time}]}$ or $\frac{\Delta P_{\text{product}}}{\Delta [\text{time}]}$) will be a positive value.
Rate of Disappearance
The reactants in a chemical reaction are being used up, so their concentration (or partial pressure) will decrease over time. The rate of disappearance of a reactant (again, still calculated with $\frac{\Delta [\text{reactant}]}{\Delta [\text{time}]}$ or $\frac{\Delta P_{\text{reactant}}}{\Delta [\text{time}]}$ will be a negative value.
Since the phrase “rate of disappearance” already shows that we know the amount of product is decreasing, a rate labelled as a disappearance may drop its negative sign for convenience. Check with your instructor or TA if they will accept this convention, or whether you should include the negative sign when reporting results.
Rate of Reaction
Compared to rate of appearance or disappearance, the rate of reaction (or “general rate of reaction”) is a special rate that has been normalized to describe one unit of “reaction” (in other words, the entire reaction as written). You can convert between rate of appearance (or disappearance) and rate of reaction using the reaction stoichiometry. See the Relative Rates of Reaction page for how to do this.
General rate of reaction is always a positive value.
Average Rates
An average rate is a rate measured across a finite time frame (e.g. from 5s to 10s or from start to completion of a reaction). You can calculate an average rate of reaction from a table of concentration or pressure data – where you know the amount of a reactant at specific times. You can also calculate an average rate graphically by taking the slope of a concentration vs time graph between two points. Both of these are demonstrated on the Determining Rates of Reaction page.
Instantaneous Rates
While an average rate is measured across a finite length of time, an instantaneous rate is measured at one exact moment. In other words, an instantaneous rate could be measured at exactly t=1s, but not from t=1 to 1.1s.
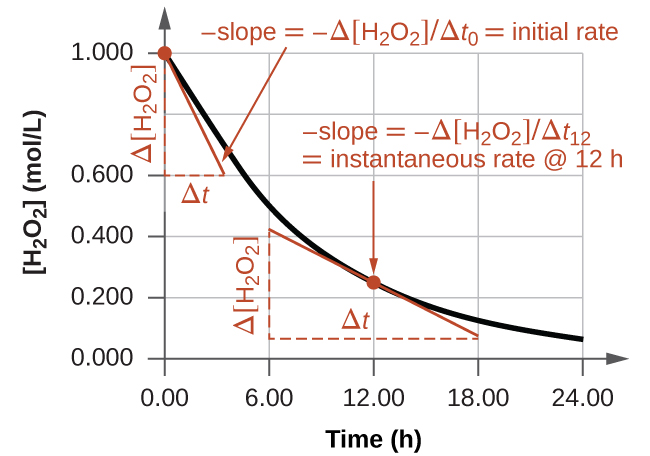
Instantaneous rates must be calculated graphically or using differentials, by finding the slope of a tangent line for a plot of concentration vs time (see the figure below). The slope of the tangent line at time t (the time you’re interested in) will be the instantaneous rate at that time.
Since you are determining the instantaneous rate from the concentration of a specific chemical, remember that it will always give you its rate of appearance (or disappearance), not the general rate of reaction.
Initial rate of reaction
The initial rate of reaction is a special type of instantaneous rate. To calculate the initial rate of reaction, take the slope of the tangent at t=0 (the initial time).