There are two ways to determine the amount of heat involved in a chemical change: measure it experimentally, or calculate it from other experimentally determined enthalpy changes. Some reactions are difficult, if not impossible, to investigate and make accurate measurements for experimentally. And even when a reaction is not hard to perform or measure, it is convenient to be able to determine the heat involved in a reaction without having to perform an experiment.
This type of calculation usually involves the use of Hess’s law, which states: If a process can be written as the sum of several stepwise processes, the enthalpy change of the total process equals the sum of the enthalpy changes of the various steps. Hess’s law is valid because enthalpy is a state function: Enthalpy changes depend only on where a chemical process starts and ends, but not on the path it takes from start to finish. For example, we can think of the reaction of carbon with oxygen to form carbon dioxide as occurring either directly or by a two-step process. The direct process is written:
$$C\;(s)+O_2\;(g)⟶CO_2\;(g)\qquad ΔH°=−394\;kJ/mol$$
In the two-step process, first carbon monoxide is formed:
$$C\;(s)+\frac{1}{2}O_2\;(g)⟶CO\;(g)\qquad ΔH°=−111\;kJ/mol$$
Then, carbon monoxide reacts further to form carbon dioxide:
$$CO\;(g)+ \frac{1}{2}O_2\;(g) ⟶CO_2\;(g)\qquad ΔH°=−283\;kJ/mol$$
The equation describing the overall reaction is the sum of these two chemical changes:
| Step 1: $C\;(s)+\frac{1}{2}O_2\;(g)⟶CO\;(g)$ |
| Step 2: $CO\;(g)+\frac{1}{2}O_2\;(g)⟶CO_2\;(g)$ |
| Sum: $C\;(s)+\frac{1}{2}O_2\;(g)+CO\;(g)+\frac{1}{2}O_2\;(g)⟶CO\;(g)+CO_2\;(g)$ |
Because the CO produced in Step 1 is consumed in Step 2, the net change is: $$C\;(s)+O_2\;(g)⟶CO_2\;(g)$$
According to Hess’s law, the enthalpy change of the reaction will equal the sum of the enthalpy changes of the steps.
$$C\;(s)+O_2\;(g)⟶CO_2\;(g)\qquad ΔH°=−111\;kJ/mol $$
$$\frac{CO\;(g)+\frac{1}{2}O_2\;(g)⟶CO_2\;(g)}{C\;(s)+O_2\;(g)⟶CO_2\;(g)}\qquad \frac{ΔH°=−283\;kJ/mol}{ΔH°=−394\;kJ/mol}$$
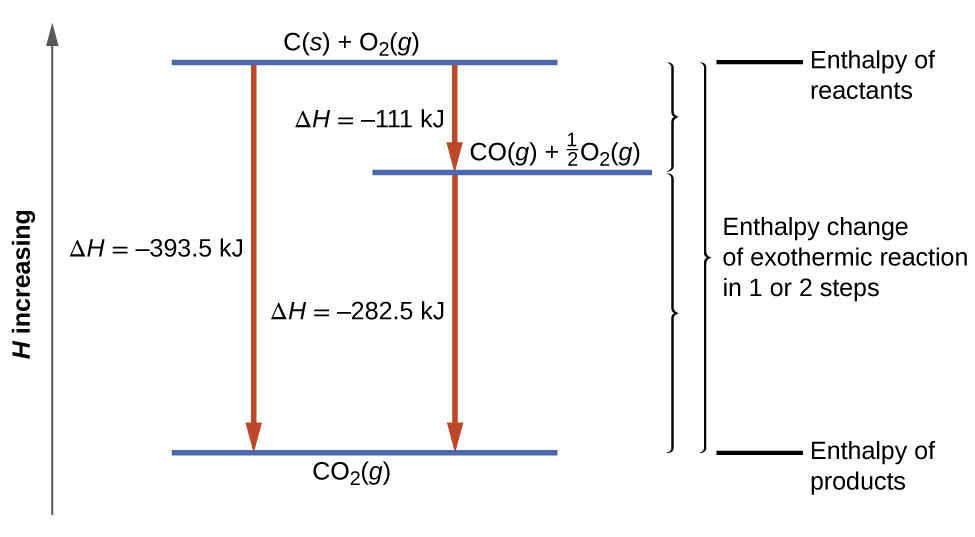
The result is shown in the figure below. We see that ΔH of the overall reaction is the same whether it occurs in one step or two. This finding (overall ΔH for the reaction = sum of ΔH values for reaction “steps” in the overall reaction) is true in general for chemical and physical processes.
Before we further practice using Hess’s law, let us recall two important features of ΔH.
- ΔH is directly proportional to the quantities of reactants or products. For example, the enthalpy change for the reaction forming 1 mole of NO2(g) is +33.2 kJ/mol: $$\frac{1}{2}N_2\;(g)+O_2\;(g)⟶NO_2\;(g)\qquad ΔH=+33.2\;kJ/mol$$ When 2 moles of NO2 (twice as much) are formed, the ΔH will be twice as large: $$N_2\;(g)+2O_2\;(g)⟶2NO_2\;(g)\qquad ΔH=+66.4\;kJ/mol$$ In general, if we multiply or divide an equation by a number, then the enthalpy change should also be multiplied or divided by the same number.
- ΔH for a reaction in one direction is equal in magnitude and opposite in sign to ΔH for the reaction in the reverse direction. For example, given that: $$H_2\;(g)+Cl_2\;(g)⟶2HCl\;(g)\qquad ΔH=−184.6\;kJ/mol$$ Then, for the “reverse” reaction, the enthalpy change is also “reversed”: $$2HCl\;(g)⟶H_2\;(g)+Cl_2\;(g)\qquad ΔH=+184.6\;kJ/mol$$
Stepwise Calculation of ΔHf° Using Hess’s Law
Determine the enthalpy of formation, $ΔH_f°$, of FeCl3(s) from the enthalpy changes of the following two-step process that occurs under standard state conditions: $$Fe(s)+Cl_2(g)⟶FeCl_2(s)\qquad ΔH°=−341.8\;kJ/mol$$ $$FeCl_2(s)+\frac{1}{2}Cl_2(g)⟶FeCl3(s)\qquad ΔH°=−57.7\;kJ/mol$$
Solution
We are trying to find the standard enthalpy of formation of FeCl3(s), which is equal to ΔH° for the reaction: $$Fe(s)+\frac{3}
Looking at the reactions, we see that the reaction for which we want to find ΔH° is the sum of the two reactions with known ΔH values, so we must sum their ΔHs:
{2}Cl_2(g)⟶FeCl_3(s)\qquad ΔH°_f=?$$| $Fe(s)+Cl_2(g)⟶FeCl_2(s)\qquad ΔH°=−341.8\;kJ/mol$ |
| $FeCl_2(s)+\frac{1}{2}Cl_2(g)⟶FeCl_3(s)\qquad ΔH°=−57.7\;kJ/mol$$ |
| $Fe(s)+\frac{3}{2}Cl_2(g)⟶FeCl_3(s)\qquad ΔH°=−399.5\;kJ/mol$ |
The enthalpy of formation, $ΔH_f°$, of FeCl3(s) is −399.5 kJ/mol.
Check Your Learning
Calculate ΔH for the process: $$N_2\;(g)+2O_2\;(g)⟶2NO_2\;(g)$$
from the following information: $$N_2\;(g)+O_2\;(g)⟶2NO\;(g)\qquad ΔH=180.5\;kJ/mol$$ $$NO\;(g)+\frac{1}{2}O_2\;(g)⟶NO_2\;(g)\qquad ΔH=−57.06\;kJ/mol$$
Answer
66.4 kJ/mol
Here is a less straightforward example that illustrates the thought process involved in solving many Hess’s law problems. It shows how we can find many standard enthalpies of formation (and other values of ΔH) if they are difficult to determine experimentally.
A More Challenging Problem Using Hess’s Law
Chlorine monofluoride can react with fluorine to form chlorine trifluoride:
(i) $ClF(g)+F_2(g)⟶ClF_3(g)\qquad ΔH°=?$
Use the reactions here to determine the ΔH° for reaction (i):
(ii)$2OF_2(g)⟶O_2(g)+2F_2(g)\qquad ΔH_{(ii)}°=−49.4\;kJ/mol$
(iii)$2ClF(g)+O_2(g)⟶Cl_2O(g)+OF_2(g)\qquad ΔH_{(iii)}°=+214.0\;kJ/mol$
(iv)$ClF_3(g)+O_2(g)⟶\frac{1}{2}Cl_2O(g)+\frac{3}{2}OF_2(g)\qquad ΔH_{(iv)}°=+236.2\;kJ/mol$
Solution
Our goal is to manipulate and combine reactions (ii), (iii), and (iv) such that they add up to reaction (i). Going from left to right in (i), we first see that ClF(g) is needed as a reactant. This can be obtained by multiplying reaction (iii) by $\frac{1}{2}$, which means that the ΔH° change is also multiplied by $\frac{1}{2}$:
$$ClF(g)+\frac{1}{2}O_2(g)⟶\frac{1}{2}Cl_2O(g)+\frac{1}{2}OF_2(g)\qquad ΔH°=\frac{1}{2}(214.0)=+107.0\;kJ/mol$$
Next, we see that F2 is also needed as a reactant. To get this, reverse and halve reaction (ii), which means that the ΔH° changes sign and is halved: $$\frac{1}{2}O_2(g)+F_2(g)⟶OF_2(g)\qquad ΔH°=+24.7\;kJ/mol$$
To get ClF3 as a product, reverse (iv), changing the sign of ΔH°: $$\frac{1}{2}Cl_2O(g)+\frac{3}{2}OF_2(g)⟶ClF_3(g)+O_2(g)\qquad ΔH°=−236.2\;kJ/mol$$
Now check to make sure that these reactions add up to the reaction we want:
Reactants $\frac{1}{2}O_2$ and $\frac{1}{2}O_2$ cancel out product O2; product $\frac{1}{2}Cl_2O$ cancels reactant $\frac{1}{2}Cl_2O$; and reactant $\frac{3}{2}OF_2$ is cancelled by products $\frac{1}{2}OF_2$ and OF2. This leaves only reactants ClF(g) and F2(g) and product ClF3(g), which are what we want. Since summing these three modified reactions yields the reaction of interest, summing the three modified ΔH° values will give the desired ΔH°: $$ΔH°=(+107.0\;kJ/mol)+(24.7\;kJ/mol)+(−236.2\;kJ/mol)=−104.5\;kJ/mol$$
| $ClF(g)+\frac{1}{2}O_2(g)⟶\frac{1}{2}Cl_2O(g)+\frac{1}{2}OF_2(g)$ | ΔH°=+107.0 kJ |
| $\frac{1}{2}O_2(g)+F_2(g)⟶OF_2(g)$ | ΔH°=+24.7 kJ |
| $\frac{1}{2}Cl_2O(g)+\frac{3}{2}OF_2(g)⟶ClF_3(g)+O_2(g)$ | ΔH°=−236.2kJ |
| $ClF(g)+F_2(g)⟶ClF_3(g)$ | ΔH°=−104.5 kJ/mol |
Check Your Learning
Aluminum chloride can be formed from its elements:
(i) $2Al(s)+3Cl_2(g)⟶2AlCl_3(s)\qquad ΔH°=?$
Use the reactions here to determine the ΔH° for reaction (i):
(ii) $HCl(g)⟶HCl(aq)\qquad ΔH_{(ii)}°=−74.8\;kJ/mol$
(iii) $H_2(g)+Cl_2(g)⟶2HCl(g)\qquad ΔH_{(iii)}°=−185\;kJ/mol$
(iv) $AlCl_3(aq)⟶AlCl_3(s)\qquad ΔH_{(iv)}°=+323\;kJ/mol$
(v) $2Al(s)+6HCl(aq)⟶2AlCl_3(aq)+3H_2(g)\qquad ΔH_{(v)}°=−1049\;kJ/mol$
Answer
−1407 kJ/mol
We also can use Hess’s law to determine the enthalpy change of any reaction if the corresponding enthalpies of formation of the reactants and products are available. The stepwise reactions we consider are: (i) decompositions of the reactants into their component elements (for which the enthalpy changes are proportional to the negative of the enthalpies of formation of the reactants), followed by (ii) re-combinations of the elements to give the products (with the enthalpy changes proportional to the enthalpies of formation of the products). The standard enthalpy change of the overall reaction is therefore equal to: (ii) the sum of the standard enthalpies of formation of all the products plus (i) the sum of the negatives of the standard enthalpies of formation of the reactants. This is usually rearranged slightly to be written as follows, with ∑ representing “the sum of” and n standing for the stoichiometric coefficients: $$ΔH° _{reaction}=∑n×ΔH_f°(products)−∑ n×ΔH_f°(reactants)$$
The following example shows in detail why this equation is valid, and how to use it to calculate the enthalpy change for a reaction of interest.
Using Hess’s Law
What is the standard enthalpy change for the reaction: $$3NO_2(g)+H_2O(l)⟶2HNO_3(aq)+NO(g)\qquad ΔH°=?$$
Solution: Using the Equation
Use the special form of Hess’s law given previously, and values from Appendix G: $$ΔH ° _{reaction}=∑ n×ΔH_f°(products)−∑ n×ΔH_f°(reactants)$$
$$=[2\require{enclose}\enclose{horizontalstrike}{\;mol\;HNO_3(aq)}×\frac{−207.4\;kJ}{\enclose{horizontalstrike}{mol\;HNO_3(aq)}}+1\enclose{horizontalstrike}{mol\;NO(g)}×+\frac{90.2\;kJ}{\enclose{horizontalstrike}{mol\;NO(g)}}]−[ 3\enclose{horizontalstrike}{mol\;NO_2(g)}×\frac{+33.2\;kJ}{\enclose{horizontalstrike}{mol\;NO_2(g)}}+1\enclose{horizontalstrike}{mol\;H_2O(l)}×\frac{−285.8\;kJ}{\enclose{horizontalstrike}{mol\;H_2O(l)}}]$$
$$=[2×(−207.4)+90.25]−[3×33.2+(−285.83)]$$
$$=–324.55+186.23$$
$$=−138.32\;kJ/mol$$
Solution: Supporting Why the General Equation Is Valid
Alternatively, we can write this reaction as the sum of the decompositions of 3NO2(g) and 1H2O(l) into their constituent elements, and the formation of 2HNO3(aq) and 1NO(g) from their constituent elements. Writing out these reactions, and noting their relationships to the $ΔH°_f$ values for these compounds (from Appendix G ), we have: $$3NO_2(g)⟶\frac{3}{2}N_2(g)+3O_2(g)\qquad ΔH°_1=−99.6\;kJ/mol$$ $$H_2O(l)⟶H_2(g)+\frac{1}{2}O_2(g)\;ΔH°_2=+285.8 kJ/mol\;[−1×ΔH°_f(H_2O)]$$ $$H_2(g)+N_2(g)+3O_2(g)⟶2HNO_3(aq)\qquad ΔH°_3=−414.8\;kJ/mol\;[ 2×ΔH°_f(HNO_3)]$$ $$\frac{1}{2}N_2(g)+\frac{1}{2}O_2(g)⟶NO(g)\qquad ΔH°_4=+90.2 kJ/mol\;[ 1×(NO)]$$
Summing these reaction equations gives the reaction we are interested in: $$3NO_2(g)+H_2O(l)⟶2HNO_3(aq)+NO(g)$$
Summing their enthalpy changes gives the value we want to determine: $$ΔH°_rxn=ΔH°_1+ΔH°_2+ΔH°_3+ΔH°_4=(−99.6\;kJ/mol)+(+285.8\;kJ/mol)+(−414.8\;kJ/mol)+(+90.2\;kJ/mol)=−138.4\;kJ/mol$$
So the standard enthalpy change for this reaction is ΔH° = −138.4 kJ/mol.
Note that this result was obtained by (1) multiplying the $ΔH°_f$ of each product by its stoichiometric coefficient and summing those values, (2) multiplying the $ΔH°_f$ of each reactant by its stoichiometric coefficient and summing those values, and then (3) subtracting the result found in (2) from the result found in (1). This is also the procedure in using the general equation, as shown.
Check Your Learning
Calculate the heat of combustion of 1 mole of ethanol, C2H5OH(l), when H2O(l) and CO2(g) are formed. Use the following enthalpies of formation: C2H5OH(l), −278 kJ/mol; H2O(l), −286 kJ/mol; and CO2(g), −394 kJ/mol.
Answer
−1368 kJ/mol