Ksp and Solubility
The Ksp of a slightly soluble ionic compound may be simply related to its measured solubility provided the dissolution process involves only dissociation and solvation, for example:
$$M_pX_q(s)⇌pM^{m+}(aq)+qX^{n-}(aq)$$
For cases such as these, one may derive Ksp values from provided solubilities, or vice-versa. Calculations of this sort are most conveniently performed using a compound’s molar solubility, measured as moles of dissolved solute per liter of saturated solution.
Calculation of Ksp from Equilibrium Concentrations
Fluorite, CaF2, is a slightly soluble solid that dissolves according to the equation:
The concentration of Ca2+ in a saturated solution of CaF2 is $2.15×10^{-4}\;M$. What is the solubility product of fluorite?
Solution
According to the stoichiometry of the dissolution equation, the fluoride ion molarity of a CaF2 solution is equal to twice its calcium ion molarity:
Substituting the ion concentrations into the Ksp expression gives
$$K_{sp}=[Ca^{2+}][F^-]=(2.15×10^{-4})(4.30×10^{-4})^2=3.98×10^{-11}$$Answer:
$$8.99×10^{-12}$$
Determination of Molar Solubility from Ksp
The Ksp of copper(I) bromide, CuBr, is $6.3×10^{-9}$. Calculate the molar solubility of copper bromide.
SolutionThe dissolution equation and solubility product expression are
$$CuBr(s)⇌Cu^+(aq)+Br^-(aq)$$ $$K_{sp}=[Cu^+][Br^-]$$Following the ICE approach to this calculation yields the table

Substituting the equilibrium concentration terms into the solubility product expression and solving for x yields
$$K_{sp}=[Cu^+][Br^-]$$ $$6.3×10^{-9}=(x)(x)=x^2$$ $$x=\sqrt{(6.3×10^{-9})}=7.9×10^{-5}\;M$$Since the dissolution stoichiometry shows one mole of copper(I) ion and one mole of bromide ion are produced for each moles of Br dissolved, the molar solubility of CuBr is $7.9×10^{-5}\;M$.
The Ksp of AgI is $1.5×10^{-16}$. Calculate the molar solubility of silver iodide.
Answer:
$1.2×10^{-8}\;M$
Determination of Molar Solubility from KspThe Ksp of calcium hydroxide, Ca(OH)2, is $1.3×10^{-6}$. Calculate the molar solubility of calcium hydroxide.
SolutionThe dissolution equation and solubility product expression are
$$Ca(OH)_2(s)⇌Ca^{2+}(aq)+2OH^-(aq)$$ $$K_{sp}=[Ca^{2+}][OH^-]^2$$The ICE table for this system is

Substituting terms for the equilibrium concentrations into the solubility product expression and solving for x gives
$$K_{sp}=[Ca^{2+}][OH^-]^2$$ $$1.3×10^{-6}=(x)(2x)^2=(x)(4x^2)=4x^3$$ $$x=\sqrt[3]{\frac{1.3×10^{-6}}{4}}=6.9×10^{-3}\;M$$As defined in the ICE table, x is the molarity of calcium ion in the saturated solution. The dissolution stoichiometry shows a 1:1 relation between moles of calcium ion in solution and moles of compound dissolved, and so, the molar solubility of Ca(OH)2 is $6.9×10^{-3}\;M$.
The Ksp of PbI2 is $1.4×10^{-8}$. Calculate the molar solubility of lead(II) iodide.
Answer:
$1.5×10^{-3}\;M$
Determination of Ksp from Gram SolubilityMany of the pigments used by artists in oil-based paints are sparingly soluble in water. For example, the solubility of the artist’s pigment chrome yellow, PbCrO4, is $4.6×10^{-6}\;$g/L. Determine the solubility product for PbCrO4.

Oil paints contain pigments that are very slightly soluble in water. In addition to chrome yellow (PbCrO4), examples include Prussian blue (Fe7(CN)18), the reddish-orange color vermilion (HgS), and green color veridian (Cr2O3). (credit: Sonny Abesamis)
Solution
Before calculating the solubility product, the provided solubility must be converted to molarity:
$$[PbCrO_4]=\frac{4.6×10^{-6}\;g\;PbCrO_4}{1\;L}\times\frac{1\;mol\;PbCrO_4}{323.2\;g\;PbCrO_4}$$ $$=\frac{1.4×10^{-8}\;mol\;PbCrO_4}{1\;L}$$ $$=1.4×10^{-8}\;M$$The dissolution equation for this compound is
$$PbCrO_4(s)⇌Pb^{2+}(aq)+CrO_4^{2-}(aq)$$The dissolution stoichiometry shows a 1:1 relation between the molar amounts of compound and its two ions, and so both [Pb2+] and $[CrO_4^{2-}]$ are equal to the molar solubility of PbCrO4:
$$[Pb^{2+}]=[CrO_4^{2-}]=1.4×10^{-8}\;M$$ $$K_{sp}=[Pb^{2+}][CrO_4^{2-}]=(1.4×10^{-8})(1.4×10^{-8})=2.0×10^{-16}$$Answer:
$1.69×10^{-4}$
Calculating the Solubility of Hg2Cl2
Calomel, Hg2Cl2, is a compound composed of the diatomic ion of mercury(I), $Hg_2^{2+}$, and chloride ions, Cl–. Although most mercury compounds are now known to be poisonous, eighteenth-century physicians used calomel as a medication. Their patients rarely suffered any mercury poisoning from the treatments because calomel has a very low solubility, as suggested by its very small Ksp:
Calculate the molar solubility of Hg2Cl2.
SolutionThe dissolution stoichiometry shows a 1:1 relation between the amount of compound dissolved and the amount of mercury(I) ions, and so the molar solubility of Hg2Cl2 is equal to the concentration of $Hg_2^{2+}$ ions
Following the ICE approach results in

Substituting the equilibrium concentration terms into the solubility product expression and solving for x gives
$$K_{sp}=[Hg_2^{2+}][Cl^-]^2$$ $$1.1×10^{-18}=(x)(2x)^2$$ $$4x^3=1.1×10^{-18}$$ $$x=\sqrt[3]{\frac{1.1×10^{-18}}{4}}=6.5×10^{-7}\;M$$ $$[Hg_2^{2+}]=6.5×10^{-7}\;M=6.5×10^{-7}\;M$$ $$[Cl^-]=2x=2(6.5×10^{-7})=1.3×10^{-6}\;M$$The dissolution stoichiometry shows the molar solubility of Hg2Cl2 is equal to $[Hg_2^{2+}]$, or $6.5×10^{-7}\;M$.
Answer:
$1.2×10^{-3}\;M$
Various types of medical imaging techniques are used to aid diagnoses of illnesses in a noninvasive manner. One such technique utilizes the ingestion of a barium compound before taking an X-ray image. A suspension of barium sulfate, a chalky powder, is ingested by the patient. Since the Ksp of barium sulfate is $2.3×10^{-8}$, very little of it dissolves as it coats the lining of the patient’s intestinal tract. Barium-coated areas of the digestive tract then appear on an X-ray as white, allowing for greater visual detail than a traditional X-ray ([link]).
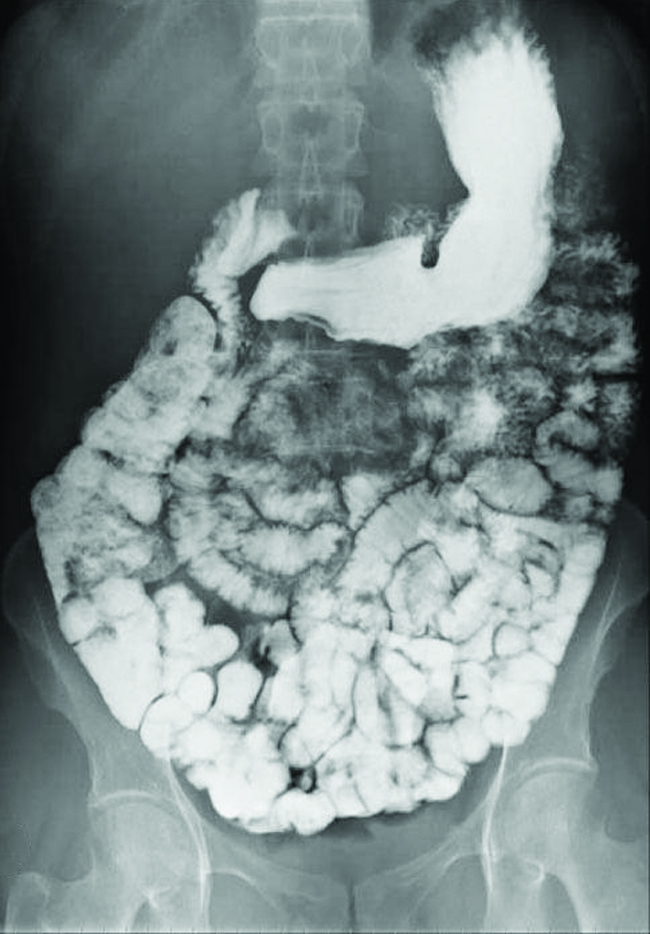
A suspension of barium sulfate coats the intestinal tract, permitting greater visual detail than a traditional X-ray. (credit modification of work by “glitzy queen00”/Wikimedia Commons)
Medical imaging using barium sulfate can be used to diagnose acid reflux disease, Crohn’s disease, and ulcers in addition to other conditions.
Visit this website for more information on how barium is used in medical diagnoses and which conditions it is used to diagnose.