Calculations with “stronger” and more dilute acids and bases
Sometimes, due to either the strength of a weak acid or base, or its concentration, its dissociation will not be negligible – and we cannot use the small-x approximation. In these cases, we simply solve the system fully, using the quadratic equation. (It is always correct to solve the system fully like this, just slower).
Calculating Equilibrium Concentrations without Simplifying Assumptions
Sodium bisulfate, NaHSO4, is used in some household cleansers as a source of the $HSO_4^-$ ion, a weak acid. What is the pH of a 0.50-M solution of $HSO_4^-$?
$$HSO_4^-(aq)+H_2O(l)⇌H_3O^+(aq)+SO_4^{2-}(aq)\qquad K_a=1.2×10^{-2}$$
Solution
The ICE table for this system is
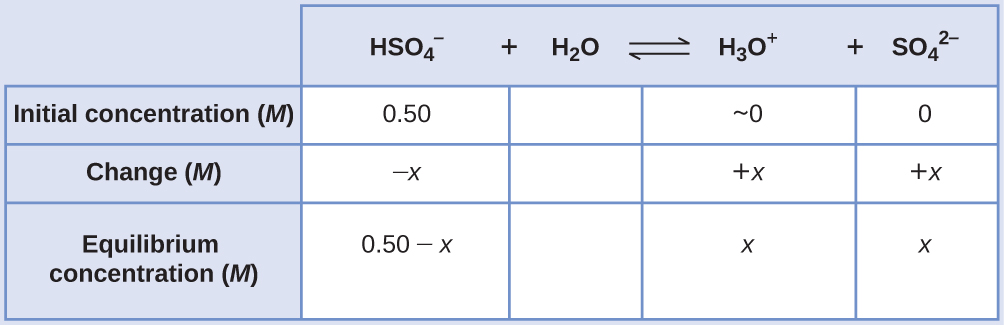
Substituting the equilibrium concentration terms into the Ka expression gives
If the assumption that x << 0.5 is made, simplifying and solving the above equation yields
This value of x is clearly not significantly less than 0.50 M; rather, it is approximately 15% of the initial concentration:
When we check the assumption, we calculate:
Because the simplifying assumption is not valid for this system, the equilibrium constant expression is solved as follows:
Rearranging this equation yields
Writing the equation in quadratic form gives
Solving for the two roots of this quadratic equation results in a negative value that may be discarded as physically irrelevant and a positive value equal to x. As defined in the ICE table, x is equal to the hydronium concentration.
Check Your Learning
Calculate the pH in a 0.010-M solution of caffeine, a weak base:
$$C_8H_{10}N_4O_2(aq)+H_2O(l)⇌C_8H_{10}N_4O_2H^+(aq)+OH^-(aq)\qquad K_b=2.5×10^{-4}$$Answer:
pH 11.16