Unlike the group 1 and 2 metal ions of the preceding examples (Na+, Ca2+, etc.), some metal ions function as acids in aqueous solutions. These ions are not just loosely solvated by water molecules when dissolved, instead they are covalently bonded to a fixed number of water molecules to yield a complex ion (see chapter on coordination chemistry). As an example, the dissolution of aluminum nitrate in water is typically represented as
$$Al(NO_3)(s)⇌Al^{3+}(aq)+3NO_3^-(aq)$$However, the aluminum(III) ion actually reacts with six water molecules to form a stable complex ion, and so the more explicit representation of the dissolution process is
$$Al(NO_3)(s)+6H_2O(l)⇌Al(H_2O)_6^{3+}(aq)+3NO_3^-(aq)$$As shown in [link], the $Al(H_2O)_6^{3+}$ ions involve bonds between a central Al atom and the O atoms of the six water molecules. Consequently, the bonded water molecules’ O–H bonds are more polar than in nonbonded water molecules, making the bonded molecules more prone to donation of a hydrogen ion:
$$Al(H_2O)_6^{3+}+H_2O(l)⇌H_3O^+(aq)+Al(H_2O)_5(OH)^{2+}(aq)\qquad K_a=1.4×10^{-5}$$The conjugate base produced by this process contains five other bonded water molecules capable of acting as acids, and so the sequential or step-wise transfer of protons is possible as depicted in few equations below:
$$Al(H_2O)_6^{3+}+H_2O(l)⇌H_3O^+(aq)+Al(H_2O)_5(OH)^{2+}(aq)$$ $$Al(H_2O)_5(OH)^{2+}(aq)+H_2O(l)⇌H_3O^+(aq)+Al(H_2O)_4(OH)_2^+(aq)$$ $$Al(H_2O)_4(OH)^+(aq)+H_2O(l)⇌H_3O^+(aq)+Al(H_2O)_3(OH)_3(aq)$$This is an example of a polyprotic acid, the topic of discussion in a later section of this chapter.
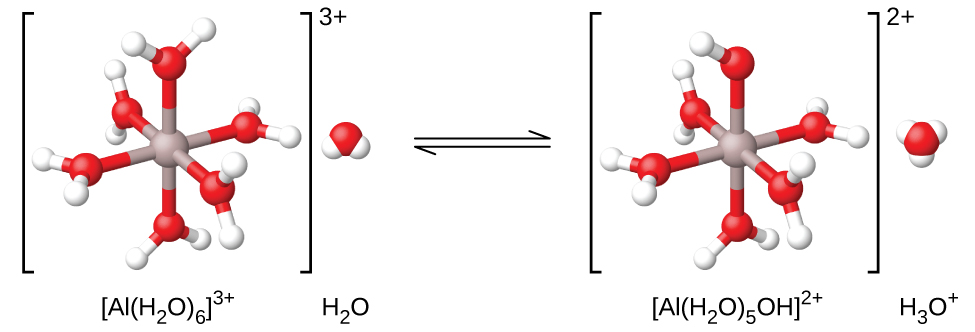
When an aluminum ion reacts with water, the hydrated aluminum ion becomes a weak acid.
Aside from the alkali metals (group 1) and some alkaline earth metals (group 2), most other metal ions will undergo acid ionization to some extent when dissolved in water. The acid strength of these complex ions typically increases with increasing charge and decreasing size of the metal ions. The first-step acid ionization equations for a few other acidic metal ions are shown below:
$$Fe(H_2O)_6^{3+}(aq)+H_2O(l)⇌H_3O^+(aq)+Fe(H_2O)_5(OH)^{2+}(aq)\qquad pK_a=2.74$$ $$Cu(H_2O)_6^{2+}(aq)+H_2O(l)⇌H_3O^+(aq)+Cu(H_2O)_5(OH)^+(aq)\qquad pK_a=~6.3$$ $$Zn(H_2O)_4^{2+}(aq)+H_2O(l)⇌H_3O^+(aq)+Zn(H_2O)_3(OH)^+(aq)\qquad pK_a=9.6$$Hydrolysis of [Al(H2O)6]3+
Calculate the pH of a 0.10-M solution of aluminum chloride, which dissolves completely to give the hydrated aluminum ion $[Al(H_2O)_6]^{3+}$ in solution.
Solution
The equation for the reaction and Ka are:
An ICE table with the provided information is

Substituting the expressions for the equilibrium concentrations into the equation for the ionization constant yields:
Assuming x << 0.10 and solving the simplified equation gives:
The ICE table defined x as equal to the hydronium ion concentration, and so the pH is calculated to be
Check Your LearningWhat is $[Al(H_2O)_5(OH)^{2+}]$ in a 0.15-M solution of Al(NO3)3 that contains enough of the strong acid HNO3 to bring [H3O+] to 0.10 M?
$2.1×10^{-5}\;M$