The kinetics chapter of this text identifies a catalyst as a substance that enables a reaction to proceed via a different mechanism with an accelerated rate. The catalyzed reaction mechanism will have a lower energy transition state than the uncatalyzed reaction, and therefore a lower activation energy, Ea, and a greater rate constant (faster reaction).
To see the effect of catalysis on an equilibrium system, consider the reaction diagram for a simple one-step (elementary) reaction shown in the figure below. The lower transition-state energy of the catalyzed reaction creates lower activation energies for both the forward and the reverse reactions. Both forward and reverse reactions will go more quickly, and equilibrium is achieved more quickly but without a change in the equilibrium constant.
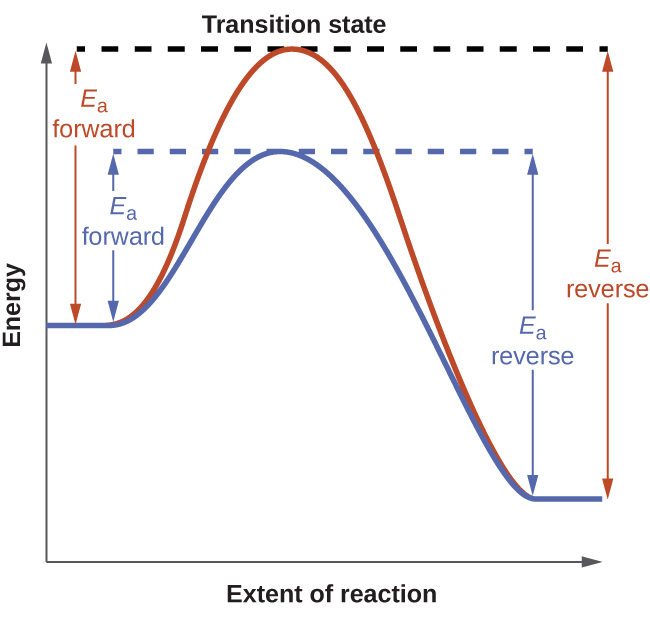
Reaction diagrams for an elementary process in the absence (red) and presence (blue) of a catalyst. The presence of catalyst lowers the activation energies of both the forward and reverse reactions but does not affect the value of the equilibrium constant.
Since adding a catalyst doesn’t change the equilibrium constant, the final (equilibrium) position of the reaction is also unchanged. Adding a catalyst won’t change the final amount of reactants or products, but you will get there faster.